![]() Contributed by Rashi Singhal, MD, MPH, and Marie DeFrances, MD, PhD
Contributed by Rashi Singhal, MD, MPH, and Marie DeFrances, MD, PhD
CLINICAL QUESTION
Among patients with hepatitis C viral infection, what role does IL28B polymorphism analysis play in predicting response to standard therapy (pegylated interferon-α and ribavirin), and in individual treatment strategy, if any?
CHIEF COMPLAINT
57-year-old African American male with chronic hepatitis C infection, deemed a nonresponder to pegylated interferon and ribavirin 9 years ago with subsequent loss to follow-up; currently presenting for clearance before evaluation for kidney transplant.
PAST MEDICAL HISTORY
SOCIAL HISTORY
Single, former laborer on disability with history of alcohol abuse 10 years ago, and 40 pack-year history of smoking. Denies use of recreational (including intravenous) drugs.
FAMILY HISTORY
Positive for diabetes, negative for cancer or liver disease.
PHYSICAL EXAMINATION
Body mass index 22 kg/m2 (weight 63 kg, height 1.7 m), in no distress, no scleral icterus, no appreciable hepatosplenomegaly, extremities without cyanosis or edema, no stigmata of chronic liver disease.
LABORATORY STUDIES
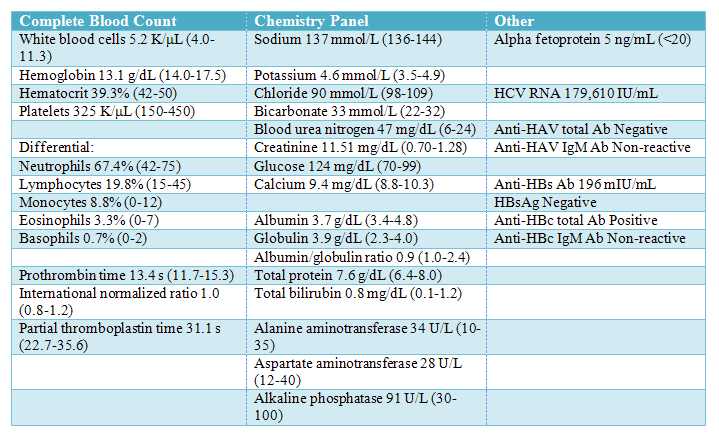
Table 1. Laboratory studies available at time of patient evaluation. Values in parentheses pertain to the laboratory reference ranges.
IMAGING STUDIES
Abdominal Ultrasound:
CLINICAL ASSESSMENT AND PLAN
Probability of sustained viral response (SVR): ~15-20% at the maximum
Recommend:
RESULTS OF ADDITIONAL STUDIES PERFORMED
Liver, Needle Biopsy*
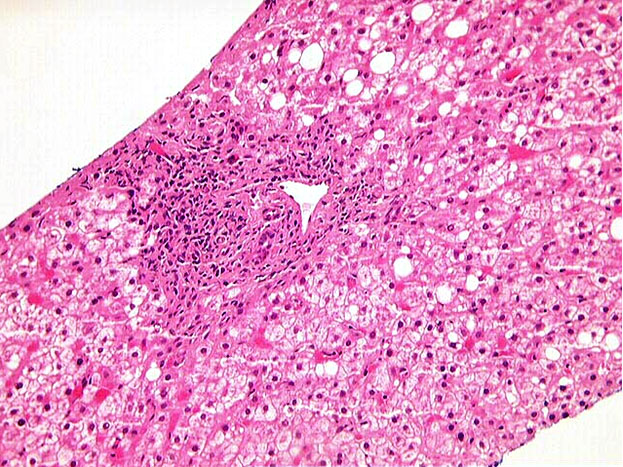
Figure 1. This low-power hematoxylin and eosin-stained liver biopsy demonstrates a periportal mononuclear infiltrate and mild periportal fibrosis. The biopsy was overall judged to show mildly active chronic hepatitis viral-type C (modified hepatic activity index 4-5/18), and mild portal fibrosis (fibrosis stage 2/6).
*A liver biopsy was performed one week after the Molecular Diagnostics laboratory received the patient's blood sample with request for IL28B polymorphism analysis, and was thus not available for review at that time. A representative image is included here for discussion, adapted from TPIS [1].
HCV Genotype Analysis
Genotype 1a
IL28B Polymorphism Analysis
Genotype CT