
![]() Contributed by Stacey Mardekian, MD and Aaron Berg, MD
Contributed by Stacey Mardekian, MD and Aaron Berg, MD
CLINICAL HISTORY
A woman in her 60s with a history of clear cell endometrial adenocarcinoma status post hysterectomy and chemotherapy, and breast cancer status post lumpectomy and radiation therapy, presented to her primary care physician with left eyelid droop. On physical exam, a nontender mass was palpated in the region of the left parotid. MRI of the brain showed no intracranial abnormality but identified an enhancing nodule in the deep portion of the left parotid gland, for which ENT evaluation was recommended.
Follow-up CT scan of the neck with contrast showed a 1.6 cm peripherally enhancing nodule in the deep lobe of the left parotid gland without evidence of extra-glandular extension but with abutment against the posterior belly of the jugulodigastric muscle. Six months later, ultrasound of the neck revealed no interval growth of the nodule, which was described as round to slightly lobulated and hypoechoic, suspicious for a primary parotid neoplasm potentially representing pleomorphic adenoma. There was no evidence of cervical lymphadenopathy on imaging.
Fine needle aspiration of the parotid mass was performed, and the patient subsequently underwent left deep lobe parotidectomy. Intraoperatively, the mass was identified as sitting just deep to the inferior branch of the facial nerve.
FINE NEEDLE ASPIRATION CYTOLOGY
Cytologic preparations showed nests of cytologically bland oncocytoid cells, characterized by uniform round nuclei and variable amounts of granular cytoplasm (Figure 1). No lymphocytes were present in the background. There was no evidence of necrosis or mitotic activity. The diagnosis was oncocytic neoplasm, with the differential diagnosis including Warthin tumor, oncocytoma, oncocytic cystadenoma and cellular pleomorphic adenoma.
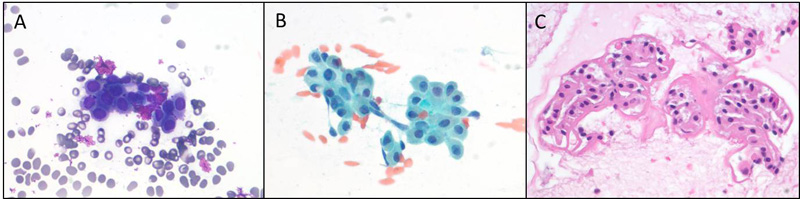
Figure 1. A, Diff-Quik stain. B, Pap stain. C, cell block.
GROSS PATHOLOGY
Gross examination of the surgical resection specimen revealed a 2.0 cm well-circumscribed uniform tan-gray firm nodule within the parotid parenchyma (Figure 2).
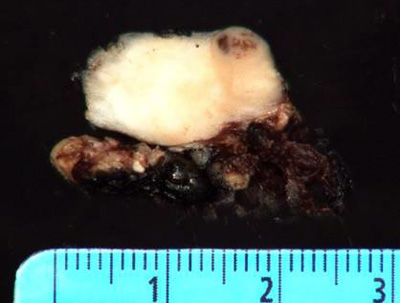
Figure 2.
MICROSCOPIC PATHOLOGY
Histologic sections showed a partially encapsulated neoplasm composed of basaloid cells with intervening islands of oncocytic cells (Figures 3A and 3B). The tumor had an infiltrative border and focally extended to the resection margin (Figure 3C). Perineural invasion was present (Figure 3D), but no angiolymphatic invasion was identified. The basaloid cells were arranged in tubular, trabecular and cribriform patterns, with intervening eosinophilic hyalinized stroma (Figures 3E, 3F and 3G).
On high power, the basaloid cells consisted of two types: a smaller cell with hyperchromatic round to oval nucleus and scant cytoplasm, and a larger more polygonal cell with pale basophilic round nucleus with nucleolus, and more ample amphophilic to slightly eosinophilic cytoplasm (Figure 3H). The larger cells occupied the center of tumor cell clusters while the smaller cells were present at the periphery, where focal nuclear palisading was evident. The basaloid tumor cells showed focal cytologic atypia and increased mitotic activity. The oncocytic cells were mostly arranged in tubules and were characterized by round nuclei and abundant granular eosinophilic cytoplasm (Figure 3I). In some areas, the basaloid and oncocytic cells could be seen intermixed (Figure 3J).
A small area of bland bilayered ductal structures embedded in chondroid matrix was also present (Figures 3K and 3L).
ANCILLARY STUDIES
By immunohistochemistry (Figure 4), CAM5.2 was positive in the larger inner tumor cells, while p63 and smooth muscle actin highlighted the outer layer of smaller tumor cells. SOX-10 was positive. S-100 was focally positive, with strongest expression in the oncocytic areas. The tumor cells showed focal nuclear positivity with beta-catenin and were positive for LEF-1. PAX-8 was negative.
Fluorescence in situ hybridization (FISH) studies for PLAG1 and MYB/NFIB translocations were negative.
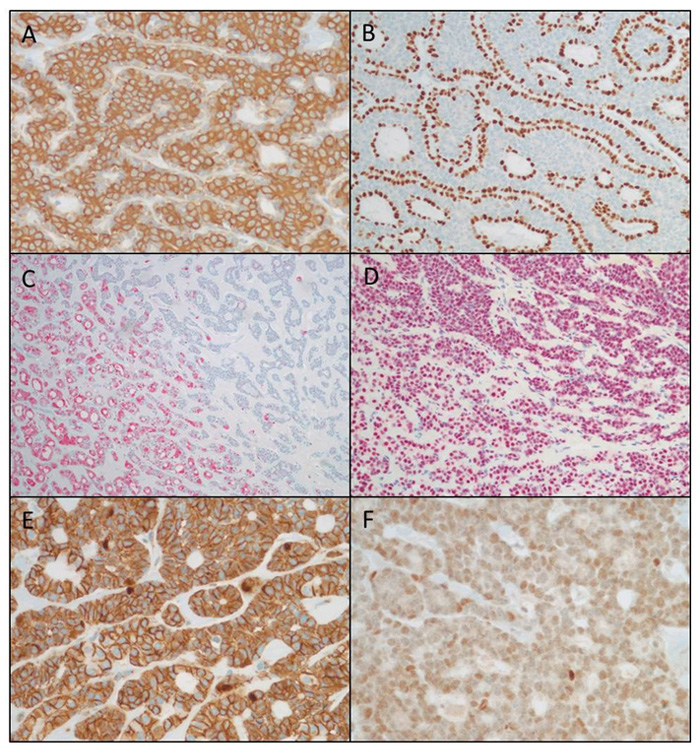
Figure 4. A, CAM5.2. B, p63. C, S-100. D, SOX-10. E, Beta-catenin. F, LEF-1.