FINAL DIAGNOSIS
SCHWANNOMA
DISCUSSION
On follow-up excision, the tumor was well-demarcated from the surrounding tissue and was able to be enucleated by the surgeon. Permanent sections demonstrated classic features confirming the diagnosis of schwannoma (discussed in detail below). Prominent palisades of round-to-spindled nuclei separated by dense, eosinophilic fibrillary processes were seen through most of the tissue (Figures 3 and 4). Regions with less cellularity and loose microcystic and myxomatous stroma were scattered throughout (figure 4). No regions of increased mitotic activity, atypical mitoses, or macronucleoli were identified.

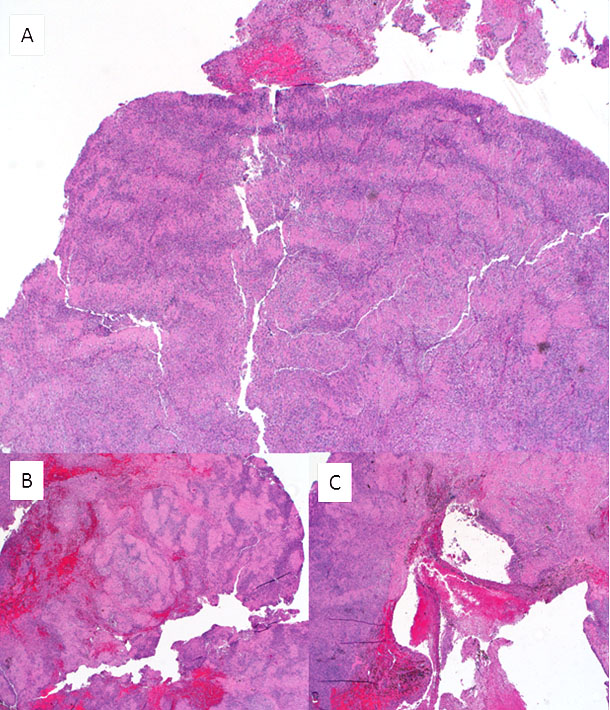
Figure 3: Excision specimen, permanent sections (H&E; low power). A-B: Alternating rows of densely palisaded nuclei and anuclear eosinophilic zones (Verocay bodies) can be seen at low power. B-C: Areas of hemorrhage, scar and hemosiderin deposition were apparent in some fragments, corresponding to the FNA site.
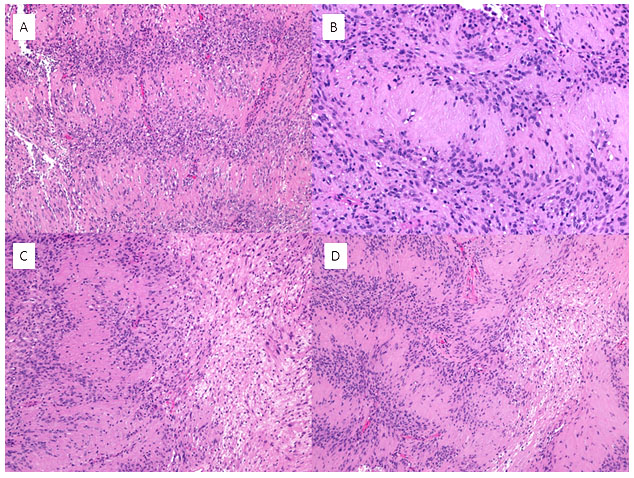
Figure 4: Excision specimen, permanent sections (H&E). Verocay bodies are seen within Antoni A areas (A-B, and left side of C and D). Looser, less cellular Antoni B areas are found immediately adjacent (right side of C and D).
Domanski et al. describe the cytologic features seen in a series of fine needle aspirates of schwannomas. [1]. The aspirates ranged from cellular and cohesive to loose, hypocellular and occasionally myxoid. Scattered isolated cells were a common finding, though very few cases (~10%) contained large quantities of dyscohesive cells. Cell morphology ranged from spindled to round, and marked pleomorphism was a diagnostic challenge in cases that ultimately were found to be "ancient schwannomas." The current case is notable not because schwannomas are rare or difficult to diagnose, but rather because the classic morphological features of this tumor were clearly apparent within the FNA specimen, a much less-common (distinctive Verocay bodies were present in <10% of the case series in [1]). Nonetheless, it was often possible to arrive at a benign diagnosis on FNA, which is important in guiding the surgical approach to excision or in allowing conservative management if clinically indicated [1].
Schwannomas, as the name suggests, are benign tumors composed of Schwann cells, the elements responsible for myelination of axons in the peripheral nervous system. Thus, schwannomas are characteristically associated with nerves, and were originally lumped with other entities into a large category of "neuromas." The defining histomorphologic features of schwannomas are eponymously named after the individuals who described them during the process of classification: in 1910 the Uruguyan neuropathologist Jose Verocay described the phenomenon of opposing rows of palisaded nuclei separated by eosinophilc, anuclear fibrillary processes now known as "Verocay bodies," and a decade later in 1920 the Swedish neurologist Nils Antoni described the characteristic mixed architectural pattern of schwannomas, with adjacent regions of cellular tissue ("Antoni A" areas, where Verocay bodies are found) and hypocellular, microcystic tissue ("Antoni B" areas) with a looser, myxomatous stroma [2]. [Note: a common misperception is that the palisaded nuclei are Antoni A areas and the anuclear fibrillary areas are Antoni B areas; this is not correct, these regions together form Verocay bodies, which are found within Antoni A areas only.] The proportion of Antoni A to Antoni B areas within a tumor can vary dramatically between individual tumors. Once these features were described and this distinctive entity could be reliably distinguished histopathologically from other nerve-associated tumors, they were renamed "neurilemmoma." It was not until ultrastructural studies by electron microscopy in the 1960s that the Schwann cell was identified as the cell of origin for the tumor, and the name "schwannoma" was coined. With the recognition that these tumors are composed of nerve sheath elements rather than neurons themselves, the more general category of "nerve sheath tumors" was adopted.
As might be expected, schwannomas can occur anywhere that Schwann cells are normally found - i.e., along peripheral nerves. That is not to say, however, that schwannomas are located exclusively in the periphery: they commonly arise intracranially along the vestibular (8th cranial) nerve root, and intradurally in the spinal column along the dorsal nerve roots, as well as in the trunk, head/neck, or extremities along peripheral nerves. Notably, bilateral "vestibular schwannomas" are one of the hallmark features of Neurofibromatosis 2 (the term "acoustic neuroma" was previously in fashion but should be retired, as it is inaccurate). Mutations in the NF2 gene, which encodes merlin, a membrane-associated tumor-suppressor protein, underlie both syndromic as well as sporadic schwannomas. The effect is overproduction of laminin, a cell-cell adhesion molecule found in the basement membrane material of Schwann cells [2]. This overexpression of laminin is hypothesized to lead to the formation of Verocay bodies.
A number of morphologic variants exist. Some schwannomas show a remarkable degree of nuclear pleomorphism and hyperchromasia, which is termed "ancient change" or degenerative atypia. Although individual cells are bizarre in morphology, they rarely contain macronucleoli or frequent mitotic figures. The most important differential is with malignant peripheral nerve sheath tumor (MPNST), which, as the name suggests, is much more aggressive clinically. MPNSTs are defined by increased mitotic activity and atypical mitoses, prominent nucleoli, and large areas of large-cell epithelioid-like change [3]. In contrast, despite having seemingly worrisome nuclear features, "ancient" schwannomas are benign. In fact, it is hypothesized that Antoni B areas arise from a degenerative process [4]. Another potentially worrisome variant is the cellular schwannoma, which consists of large stretches of cellular areas with spindled cells often arranged in a storiform pattern. The typical morphologic features can often be seen focally however, and S100 immunohistochemistry is characteristic. Melanotic schwannomas can raise the possibility of melanoma, and can be seen in the context of Carney complex. Other peripheral nerve sheath tumors such as neurofibroma, perineurioma or traumatic neuroma can be excluded by the lack of axonal elements within the tumor or the lack of strong S100 staining by immunohistochemistry [4]. Other tumors in the differential are ones that show palisading, such as palisaded leiomyoma or gastrointestinal stromal tumor (GIST) [3], though these also are excluded by strong S100 staining.
REFERENCES
![]() Contributed by Thomas Pearce, MD, PhD and Juan Xing, MD
Contributed by Thomas Pearce, MD, PhD and Juan Xing, MD