DISCUSSION
Troponin Testing for Myocardial Infarction:
The World Health Organization had defined the diagnostic criteria for myocardial infarction to include an increase in serial measurements of cardiac enzymes, electrocardiographic changes and clinical symptoms of chest pain (4). The 2007 American College of Cardiology (ACC) definition of Myocardial infarction includes: detection of rise and/or fall of cardiac biomarkers with at least one value above the 99th percentile of the upper reference limit with evidence of myocardial ischemia. Myocardial ischemia includes one of the following: symptoms of ischemia, ECG changes indicative of new ischemia (new ST-T changes or new left bundle branch block (LBBB)), development of pathological Q waves on ECG or imaging evidence of new loss of viable myocardium or new regional wall motion abnormality (3).
Concerning serial Troponin I determination, the ACC 2007 recommendations state that Troponin I or T values should be drawn on first assessment and 6-9 hours later with an additional level 12-24 hours later if the initial values were not elevated but the clinical suspicion is high for myocardial infarction. One elevated Troponin above the decision level (greater than the 99th percentile, or greater than 0.04 ng/mL at UPMC Presbyterian) is required to establish the diagnosis of myocardial infarction. The trend established by subsequent Troponin values helps rule out continued infarction, re-infarction, as well as conditions that may give rise to falsely elevated Troponin levels such as renal failure (3).
Troponins and Other Laboratory Values for Myocardial Infarction:
Troponin: There are three subunits of Troponin: Troponin Inhibitory unit (TnI), Troponin Tropomyosin binding unit (TnT) and Troponin Calcium binding unit (TnC). In muscle contraction, calcium is transmitted along Troponin which creates a conformation change in tropomysin and allows actin and myosin to interact resulting in contraction. So why do we use TnI for determination of myocardial damage?
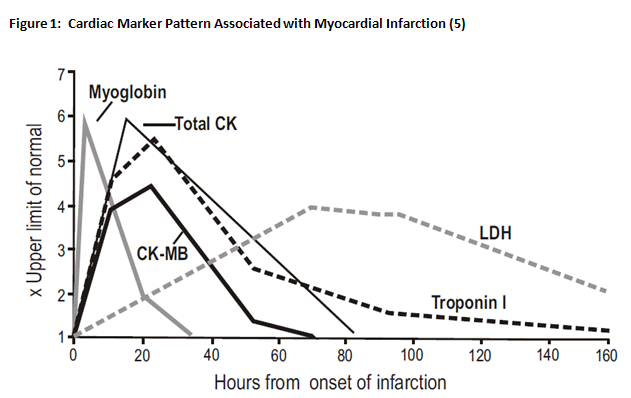
TnI is encoded by different genes in cardiac and skeletal muscle. Cardiac TnI (cTnI) contains an additional 31 amino acid residues on the N-Terminus. TnI elevation occurs 4-8 hours after chest pain onset, peaks at 12-16 hours and remains elevated for 5-9 days. This is summarized in Figure 1 (6). TnC forms found in skeletal muscle and cardiac muscle are identical, so it cannot be used as a marker to target cardiac damage. TnT does have two different forms, one in cardiac and one in skeletal muscles. Assays use antibodies specific for cardiac TnT (cTnT); however, the cTnT has been shown to be upregulated in conditions such as muscular dystrophy and renal failure.
The detection limit of cardiac Troponin (cTn) levels for most assays is 0.01 ng/mL. The 99th percentile for cTn in a normal population is reported as 0.04 ng/mL; however cTn elevations have been reported in other conditions, such as: myocarditis, pericarditis, pulmonary embolism and sepsis. Additionally, newborns can have levels as high as 3.0 ng/mL. (1)
Creatine Kinase MB (CK-MB): Two chains (M and B) form three isozymes CK-MB, CK-MM and CK-BB. CK-MM and CK-MB are found in skeletal muscle, so skeletal muscle disease will be associated with an elevated CK-MM and CK-MB. CK-MB will rise 4-6 hours after symptom onset, peak around 12 hours and Progressive CK-MB determination showing an elevation followed by a decrease is indicative for AMI. (1) This is summarized in Figure 1 (5).
Other biomarkers of Myocardial Infarction that are no longer used or have uncertain clinical relevance include: lactate dehydrogenase, myoglobin, carbonic anhydrase, glycogen phosphorylase, heart fatty acid binding protein, ischemia modified albumin (1).

Troponin Assay at UPMC Presbyterian vs UPMC Magee Women's Hospital:
UPMC Presbyterian:
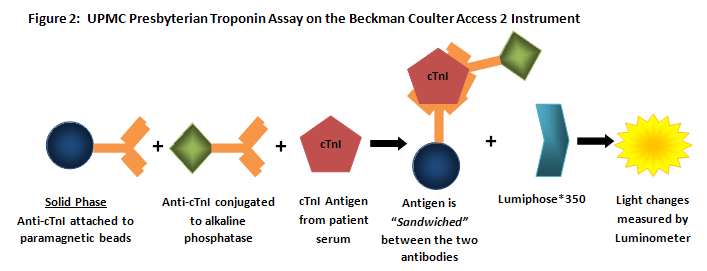
The cTnI assay at UPMC Presbyterian is run on the Beckman Coulter Access 2 which is not part of the Beckman Coulter DXC/DXI automation platform. This is a heterogeneous noncompetitive sandwich assay in which anti-cTnI antibody with specificity for one cTnI epitope is conjugated to paramagnetic beads (solid phase) with an anti-cTnI antibody with specificity for another epitope conjugated to alkaline phosphatase (ALP) is mixed with patient serum with suspected cTnI. After conjugation, Lumi-Phos*350 is added which reacts with the ALP to produce light which is measured with a luminometer and is directly proportional to the concentration (6). The principle of this assay is summarized in Figure 2.
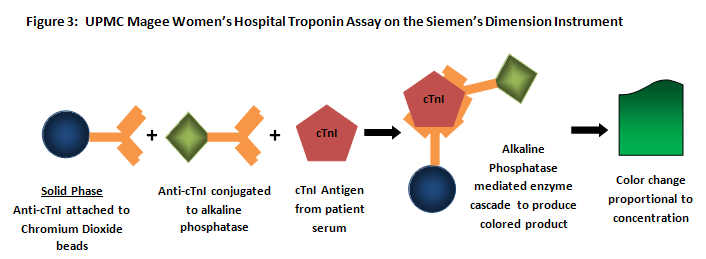
UPMC Magee Women's Hospital Troponin Assay:
The cTnI assay is run on the Siemen's Dimension platform. This is, again, a heterogenous noncompetitive assay which relies on cTnI antibodies bound to ALP and Chromium Dioxide beads to create a sandwich with the cTnI antigen. After washing, the ALP reacts with synthetic Flavin Adenine Dinucleotide Phospate (FADP) to produce FAD. FAD binds to apo D-amino acid oxidase which is converted to active holo D-amino acid oxidase which produces Hydrogen Peroxide (H2O2). The H2O2 in the presence of horseradish peroxidase (HRP) converts 3,5-dichloro-20hydroxybenzenesulfonic acid (DCHBS) and 4-aminoantipyrine (4-AAP) to a colored product that absorbs light at 510 nm (7). The principle of this assay is summarized in Figure 3.
REFERENCES
![]() Contributed by Brian K. Theisen, MD and Octavia M. Peck-Palmer, PhD
Contributed by Brian K. Theisen, MD and Octavia M. Peck-Palmer, PhD