
![]() Contributed by Lin Liu, MD, PhD and Sara Monaghan, MD
Contributed by Lin Liu, MD, PhD and Sara Monaghan, MD
CLINICAL HISTORY
The patient is a 30-year old man with a 30-pound weight loss in one month, poor appetite, and episodes of abdominal and back pain. He has had progressive pancytopenia over 1 month.
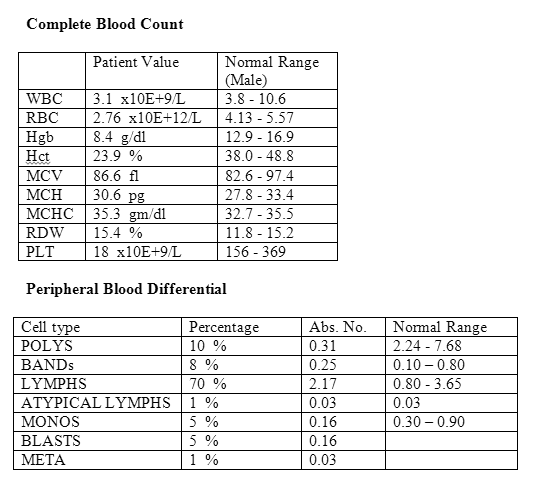
PERIPHERAL BLOOD FILM
RED BLOOD CELL MORPHOLOGY: Mild anisocytosis, mild polychromasia, and rare nucleated red blood cells (Figure 1).
WHITE BLOOD CELL MORPHOLOGY: Blasts are medium to large with scant to moderate cytoplasm, fine chromatin, round to irregular nuclei, and small nucleoli. Some blasts display fine granules, and some appear monocytic and could be classified as promonocytes (Figure 2).
PLATELETS: Decreased.
BONE MARROW
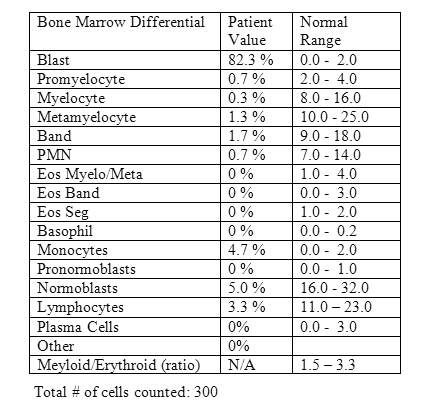
The marrow aspirate smears are paucispicular with some necrotic material, but display intact cells in some areas (Figures 3 and 4). The bone marrow biopsy is approaching 100% cellular in some areas, but also shows large areas of coagulative necrosis (Figures 5 and 6). The aspirate smears and intact areas on the biopsy display numerous blasts with a similar morphology to those observed in the peripheral blood (Figures 3, 4 and 7). Normal hematopoiesis is almost completely effaced. Only very rare maturing granulocytes and erythroid precursors are seen. Megakaryocytes appear absent. No ringed sideroblasts are identified on the iron stain done on a touch imprint, but erythroid precursors are rare.
ANCILLARY STUDIES
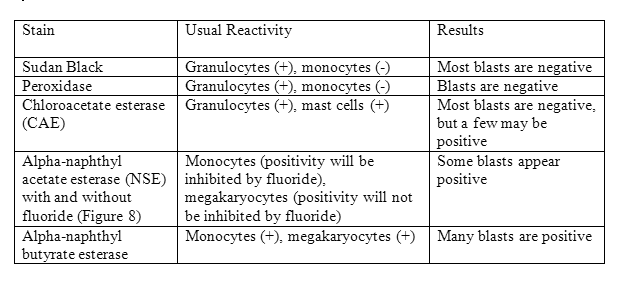
CYTOCHEMICAL STAINS
Cytochemical stains were performed on the aspirate smears or touch imprints. The following results were found:

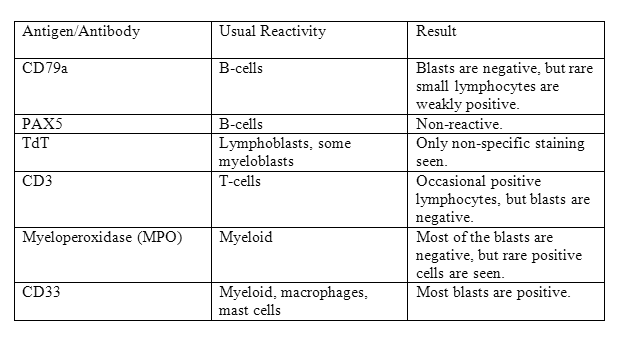
In order to further characterize the immunophenotype of the blasts, paraffin section immunohistochemical studies were performed on bone marrow biopsy. The following results were found:
FLOW CYTOMETRY (Figures 11 and 12)
The flow cytometric immunophenotypic studies performed on the bone marrow demonstrate an abnormal myeloid population that shows evidence of monocytic differentiation, comprising about 77% of the total cellular events. This population has the following immunophenotype: dim CD45 positive, CD34 negative, CD117 negative, CD13 predominantly negative, CD33 bright positive, CD15 variably positive, CD64 positive, HLA-DR positive, CD36 partial positive, and CD11b variably positive. B and T-lymphoid markers are not assessed due to the low cellularity. Also present are occasional lymphocytes that are not further characterized due to the lack of sufficient sample.
CYTOGENETIC STUDIES
52,XY,+3,+6,+7,+7,+9,+18[cp3]/46,XY[2]
nuc ish(ABL1x2~3,BCRx2)[274]
nuc ish(MLLx2)[201]
Mosaic abnormal male bone marrow chromosome analysis with an apparently normal cell line and one that is hyperdiploid with 52 chromosomes, including trisomies 3 and 6, tetrasomy 7 and trisomies 9 and 18. Hyperdiploidy with more than 48 chromosomes is a relatively rare finding in AML and is not associated with a particular AML subtype or prognosis [Kohler, M, et al. (1994) Cancer Genet Cytogenet 73:79-81]. There were no structural abnormalities found in this specimen. However, the number of mitotic cells was less than optimal (five instead of the usual 20), reducing the likelihood of detecting the putative additional chromosome aberrations. Fluorescence in situ hybridization (FISH) was negative for the BCR/ABL1 and MLL gene rearrangements in all 274 and 201 interphase cells examined for each test, respectively. An extra signal for ABL1 (9q34) in 73% of the interphase cells examined for corresponds to trisomy 9 detected by the concurrent classical chromosome analysis.
Classical cytogenetic analysis on the metaphase cells and FISH for the PML/RARA gene rearrangement on the interphase cells from a subsequent unstimulated peripheral blood specimen is in progress and will be reported when complete.