FINAL DIAGNOSIS: NONSECRETORY MULTIPLE MYELOMA (NSMM)
BACKGROUND:
Although multiple myeloma is one of the most common malignancies in the United States, NSMM accounts only for approximately 1% of these cases. The patient may present with a range of severity of symptoms including fatigue, skeletal destruction with osteolytic lesions, pathological fractures, bone pain, hypercalcemia and anemia. Until recently, this neoplasm was described as synthesizing, but not secreting, immunoglobulin molecules which led to an absence of an M-component on serum protein electrophoresis and/or urine protein electrophoresis, as well as immunofixation. The WHO diagnostic criteria for multiple myeloma are as follows:
Major criteria (3)
Minor criteria (4)
Diagnostic requirements: The diagnosis of multiple myeloma requires a minimum of one major criterion and one minor criterion, or three minor criteria which must include bone marrow plasmacytosis of 10-30 percent and the presence of a monoclonal protein. These criteria must be manifest in a symptomatic patient with progressive disease. 1
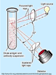
FREE LIGHT CHAIN (FLC) ASSAY:
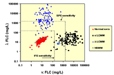
The FLC assay is performed by nephelometry (Figure 8). The antibodies used are specific for the portion of the light chain which would not be exposed if it were attached to an immunoglobulin. This assay is able to detect light chains below the sensitivity of serum/urine protein electrophoresis and immunofixation (Figure 9).5,6 This also allows, not only the detection of NSMMs, but also the more accurate measurement of Light Chain Multiple Myeloma (LCMM) in the serum, rather than looking at the Bence Jones proteins in the urine (since the kidneys metabolize large amounts of free light chains).2,7
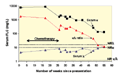
The amount of κ and λ light chains and their ratio can now be followed in order to monitor the patient's response to treatment and disease status without invasive bone marrow biopsy, as seen in the example (not from the case patient) (Figure 10).5
DISCUSSION:
In this case, the patient's biopsy satisfies WHO diagnostic requirements, as well as allowing the identification of the κ light chan restricted plasma cell clone by flow cytometry and immunohistochemistry. The serum protein electrophoresis and the immunofixation showed no M-component or κ light chain. However, when an sFLC assay of the patient's serum was performed, the amount of κ light chains, as well as the κ/λ ratio were markedly increased, demonstrating the light chain restricted clone by a less invasive test. The sFLC assay may now be used to monitor the patient's progress as he undergoes treatment.
Acknowledgment: The author would like to thank Dr. Rabin and Dr. Roodman for their suggestions and guidance.
REFERENCES:
![]() Contributed by Marian Rollins-Raval, MD, MPH
Contributed by Marian Rollins-Raval, MD, MPH