FINAL DIAGNOSIS:
DISCUSSION:
The original tumor from 1992 was reviewed and immunohistochemically evaluated confirming the original diagnosis, but quite different from the current tumor (tumor cells were synaptophysin, chromogranin, CAM5.2, LCA and neurofilament protein immunonegative; marked cytoplasmic GFAP immunopositivity was present). In 2002, however, the tumor showed sheets of densely packed, small dark cells with a scanty cytoplasm expressing NCAM and synaptophysin with only focal GFAP expression. This dramatic change in morphology was considered a second tumor induced by previous irradiation. However the possibility of progression of the original tumor (modulated by radiation effect) was not excluded in view of GFAP-immunopositive cells in the current tumor, albeit in a minority.
Radiation induced cerebral neoplasia is a well known phenomenon [7]. White matter is more vulnerable to radiation damage than grey matter [8] especially in doses greater than 20 Gy. The evolution of these new tumor entities after irradiation ranges 5-10 years in adolescence and adults [2-4] [6] and few years only in children [5]. Radiation induced long-term side effects for benign brain tumors are well documented [1,9]. Speculation on the biological nature of secondary tumors are fascinating [9]. Not well established is the relation between irradiation of low grade glioma and consecutive emergence of primitive neuroectodermal tumor (PNET).
Activation of K-ras gene has been implicated in the emergence of a (PNET) after radiation therapy for acute lymphocytic leukemia [3]. There is also growing evidence for emergence of PNET after radiation therapy in gliomas [5,9].
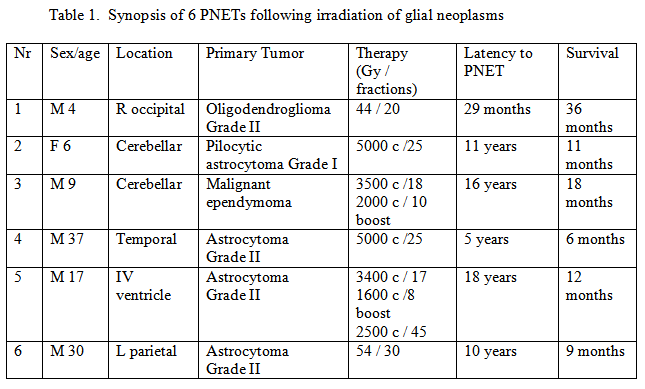
To the 5 cases reported so far we add a 6th case. The present case shows the emergence of a supratentorial PNET after irradiation for an astrocytoma, WHO grade II. Table 1 shows a synopsis of all reported PNET cases after irradiation for a primary glial neoplasm. In all cases total removal of the primary neoplasm was achieved (patient 4 is reported to had a subtotal resection).

In patients 1, 5 and 6 the PNET appeared at the site of the irradiated primary lesion. The first patient had a recurrence of the primary tumor after 9 months. He developed a PNET after 29 months from the 1st operation. It appears the survival period roughly equals the number of years of latency to PNET expressed in months, although this needs further cytogenetic and clinical examination.
Occurrence of PNET after irradiation of primary brain tumor appear to be more common than previously thought. Evolution into these "secondary PNETs" needs to be considered in patients receiving radiotherapy for a glial neoplasm. It also adds to the differential diagnosis in patients with a long-term sequela of irradiation. Table 1 shows the aggressive nature of these "secondary PNETs". In the majority of cases, the survival time appears shorter than known for supratentorial PNETs [10].
REFERENCES
![]() Contributed by Atik Baborie , Arundhati Chakrabarty, Sarin Kuruvath, Jake Timothy , Graham Bonsor , Phillip van Hille, Leslie R. Bridges
Contributed by Atik Baborie , Arundhati Chakrabarty, Sarin Kuruvath, Jake Timothy , Graham Bonsor , Phillip van Hille, Leslie R. Bridges