
![]() Contributed by Ming Yin, MD, PhD and Mona F. Melhem, MD
Contributed by Ming Yin, MD, PhD and Mona F. Melhem, MD
![]() Published on line in April 2006
Published on line in April 2006
PATIENT HISTORY:
The patient is a 74-year-old Caucasian male, who had intermittent skin rash for seven years. Rash usually goes away with medicated creams, but recurs every year. A skin biopsy was obtained at a local hospital four years ago and was suggestive of mycosis fungoides (cutaneous T-cell lymphoma). He was treated with oral and topical steroids. The topical therapy included prednisone (with a taper), triamcinolone and fluocinonide. In the past 2 years, the patient was found to have anemia (hemoglobin was 10.3 g/dl) with a normal white cell count and thrombocytopenia (96 x 109/L) of unknown etiology. Laboratory studies indicated normal values for reticulocyte count, serum levels of B12, folate, ferritin, and total iron binding capacity. He did have a history of heme-positive stool, but refused colonoscopy. The patient underwent a bone marrow biopsy one and half years ago. The results included a hypercellular marrow with trilineage hematopoiesis, focally increased and dysplastic megakaryocytes, suggestive of myelodysplastic syndrome (MDS) and no increase in blasts. Flow cytometry immunophenotyping did not identify an aberrant T or B cell immunophenotype. Cytogenetic studies were normal. He had been treated with Procrit.
Four months ago, the patient presented with increased rash and itching. On physical examination, patchy erythematous papules and dry, scaling plaques were seen over his whole right face (forehead, eye area, cheek, jawline) extending over the midface area to left infraorbital and left cheek. A similar skin rash was also found in bilateral upper and lower extremities, sparing the palms and the feet. A skin punch biopsy was performed in the neck area.
The skin biopsy revealed a dense perivascular and epidermotropic lymphocytic infiltrate composed of small to medium-sized, lymphocytes with round to irregular nuclei, and scattered eosinophils and plasma cells (figure 1A). There was a Pautrier microabscess within epidermis (figure 1B). Immunohistochemical stains showed strong reactivity for T-cell markers: CD3, CD4 (figures 1C, 1D and 1E), and CD2. Only scattered CD20 positive B-cells were present. The findings were consistent with cutaneous T-cell lymphoma (CTCL), early plaque stage.
He was treated with prednisone, triamcinolone, and 0.05% Lidex (topical corticosteroid). One month later, the skin lesion of CTCL was seen involving nearly 95% of his body surface, with areas of oozing a clear, sticky fluid. The patient also showed early "lion-like changes" of the central face. One month prior to the admission, the patient complained of fever with chills, loss of appetite and decreased body weight. Peripheral smear revealed an increased number of white blood cells with "atypical lymphocytes" and increased "immature mononuclear cells". Based on his clinical presentations, the possibility of CTCL exacerbation with Sezary's syndrome was suspected and a full hematologic analysis was carried out.
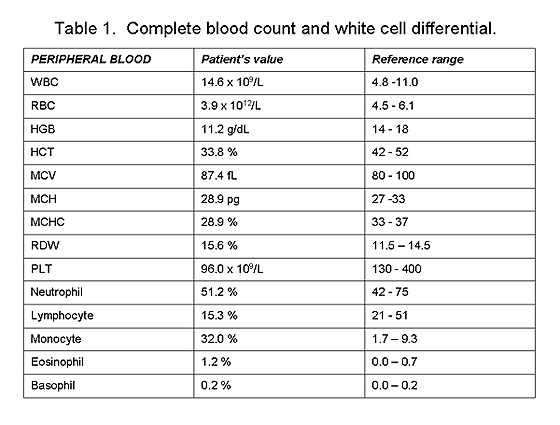
The complete blood count and white cell differential showed mildly increased white blood cell count (14.6 K/cmm), with markedly increased number of monocytes and reduced number of lymphocytes (table 1). The peripheral smear revealed an increased number of atypical monocytes with cytoplasmic vacuoles (figures 2A and 2B). Hypogranular neutrophils and mature lymphocytes were also seen.
Flow cytometry immunophenotypic studies performed on bone marrow revealed heterogeneous cellular populations, including granulocytes, monocytes and lymphocytes (figures 3A, 3B, 3C, 3D and 3E). The population of CD14+ monocytes was increased, comprising about 15% of total cells. The granulocytes showed reduced side-scatter light characteristics, suggested of hypogranularity. The population of CD34+ blasts (0.4% of total cells) and CD117+ cells (1.5% of total cells) were not increased. T-lymphocytes comprised about 3-3.5% of total cells and showed no overt immunotypic evidence of aberrancy. There were also rare polyclonal B lymphocytes and NK cells. Bone marrow aspirate (figure 4) demonstrated a myeloid predominance with a M:E ratio of 4.1:1. The erythoid and myeloid series displayed marked nuclear cytoplasmic asynchrony. There was an increase in monoblasts (23%) and mature monocytes (11%). Megakaryocytes were present. Bone marrow biopsy was 80% cellular. There were clusters of immature cells scattered throughout the marrow spaces (figure 5A). These cells were large with convoluted nuclei and occasional prominent nucleoli, consistent with monoblasts and monocytes that were positive for CD68 (figure 5B) and negative for CD20, CD3, CD30, S100 and pancytokeratin. Occasional maturing myeloid cells were present. Megakaryocytes were decreased with 0-2 per high power field, some appear to be dysplastic. Iron stores were markedly decreased to absent.
Cytogenetic study on bone marrow reveals 46, XY.