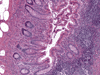


FINAL DIAGNOSIS:
There are two separate diagnoses in this case:
DISCUSSION:
Patients with HIV infection may be immunosuppressed and therefore susceptible to opportunistic infections. This patient presented with diarrhea, weight loss, and abdominal pain. The main cause of his symptoms is likely the CMV infection, which can cause mucosal ulceration, abdominal pain and bloody diarrhea. Although no viral inclusions were readily identified on the H&E stain, in a patient with colonic ulcers and a history of HIV infection, a CMV immunostain can help identify the organisms within the tissue. However, this patient had more than one infection, which is an important point to this case presentation. Once one infectious organism is found in a patient with HIV/AIDS, other infections, including fungal, mycobacterial (i.e. Mycobacterium avian intercellulare), bacterial and viral, should be excluded either histologically, cytochemically or immunohistochemically. In this case, this patient also had intestinal spirochetosis, which causes watery diarrhea, one of this patient's presenting symptoms.
Intestinal Spirochetes
Background:
The presence of spirochetes in the gut flora was first described in 1884. Although spirochetes have been noted in human feces since the end of the last century, the attachment of spirochetes to the surface of colonic mucosa forming a "false brush border" was first described in1967 (1). It was referred to as intestinal spirochetosis (IS). This condition can affect both adults and children.
Debate exists as to whether the spirochetes are true pathogens. Intestinal spirochetes may represent enteric commensals that become opportunistic due to alteration in the local environment. Some patients have been reported to present with abdominal pain, diarrhea, constipation and rectal bleeding (2). Appendiceal spirochetosis has been found in patients with histologically proven appendicitis, in patients with pseudoappendicitis or in asymptomatic patients (3) (Figures 9, 10 and 11 [H&E and Steiner stains] are from an appendectomy specimen performed for presumed acute appendicitis; no acute appendicitis was noted, but spirochetes were identified). Antibiotic treatment with metronidazole can improve symptoms, but some patients may not respond or may improve spontaneously without therapy (4). The fact that spirochetes have been demonstrated in the stool, colon and appendix, and in a wide variety of intestinal diseases, including carcinoma of the colon, adenomas, ulcerative colitis, diverticulitis, Crohn's disease and hemorrhoids, further suggests that intestinal spirochetes may be non-pathogenic commensals. On the other hand, IS has been noted as a cause of diarrhea in pigs and dogs, and experimental infections of pigs and birds with human isolates of this organism have led to intestinal disease strongly suggesting that these organisms might in fact have pathogenic potential.
The disease has a worldwide distribution with differing rates of prevalence. Prevalence rates of IS are higher in the developing world than developed countries, ranging from 11.4 to 64.3% in developing countries (5). The prevalence of IS among adults in the United States and Europe ranges between 4.5 and 32.2% (6). By contrast, infection with one spirochete, B. pilosicoli, is endemic among villagers in Papua New Guinea; 93.6% of the population is infected (7-8). Histological studies have suggested that spirochetes are present in 2.5 to 9% of rectal biopsy specimens from unselected Europeans (9-10), but are more common (64%), in biopsy specimens from individuals in southern India (11). While some studies have found no sex or age predilection for infection with IS (8-9), a recent retrospective study of 14 cases of colonic spirochetosis demonstrated that colonic spirochetosis had a male predominance and tended to occur in two distinct clinical settings: children who often had abdominal symptoms and adults who typically were asymptomatic (12).
The mode of spirochetal transmission is unknown. The differences in prevalence rates now and 100 years ago, as well as between developed and developing areas of today's world, suggest that transmission is modified by a variety of factors including diet, sanitation and community structures (4). There appears to be no association (no increased incidence) with HIV disease (13); however, patients with HIV/AIDS tend to be symptomatic from the spirochete infection (12). The high frequency of coinfections with other organisms makes it difficult to determine the clinical significance of IS in this group. In particular, infections with Neisseria gonorrhea, Entamoeba histolytica and Treponema pallidum have been seen commonly with IS (14). Interestingly, in the western world, a population at risk of infection is homosexual men with prevalence rates ranging from 20.6-62.5% (4,14-16). This suggests that intestinal spirochetes may also be sexually transmitted.
Pathologic findings:
Spirochetes have been identified in the colon and appendix. IS tends not to produce any gross or endoscopic lesions and is thus a histological diagnosis. Colorectal biopsies in patients with IS reveal a subtle thickening or accentuation of the colonic brush border, which appears deeply stained with hematoxylin (a "fuzzy" or "carpeted" mucosal surface). The organisms are found diffusely along the surface mucosa with only superficial extension into the crypts. The organisms tend to spare the goblet cells. IS is not strongly associated with active inflammation, mucosal injury, or changes of chronicity (12). A local immune response in the form of an increase in immunoglobulin E-producing plasma cells has been described in one study (17). Identification of the spirochetes can be enhanced by use of silver stains such as Warthin-Starry, Churukian-Schenk and the Steiner and Steiner stain. The organisms also stain less intensely with Giemsa, periodic acid-Schiff and Alcian blue at pH 2.5. Immunohistochemistry using a polyclonal spirochete antibody may facilitate the diagnosis but generally is not very helpful (18). Ultrastructural studies show spirochetes aligned in parallel and embedded in the luminal border of the absorptive cells. There is blunting or loss of normal microvilli. Rarely, spirochetes may be found within the cytoplasm of the epithelial cell or within macrophages in the lamina propria. Mechanisms of invasion are unknown and invasion is not always associated with clinical disease.
Microbiology:
Intestinal spirochetes infect humans and various animal species such as swine, dogs, rats, mice, chicken and opossum (19). They comprise a heterogeneous but related group of bacteria (20). Two distinct forms can colonize humans. One is Brachyspira aalborgi, which was first identified in 1982 in Denmark (21) and is the major causative agent of IS in the US (12). It is a gram-negative motile heliciodal bacterium with tapered ends 1.7-6�m in length and 0.2�m in diameter and grows in anaerobic cultures after at least 2 weeks of incubation. Culture medium comprises of 5-10% CO2 at 37oC on trypticase soy agar plates with 5-10% blood (22). The organisms are also weakly ?-hemolytic.
Soon after characterization of B. aalborgi, it became evident that a substantial number of human intestinal spirochetes were distinct from B. aalborgi and resembled porcine pathogenic and non-pathogenic strains both morphologically and by culture characteristics. This second group of spirochetes was recognized to be genetically different from Serpulina hyodysenteriae, the pathogen of swine dysentery, but identical to a pathogen isolated from pigs, Serpulina pilosicoli. It has a length of 4-12�m with more coiled and tapered ends as well as faster growth in anaerobic cultures. Colonies are normally identified after 6 days of incubation. It has been suggested that the genus Serpulina be unified with Brachyspira because of sufficient genetic and biochemical similarities. Therefore, some authors refer to Serpulina pilosicoli as Brachyspira pilosicoli. Organisms cannot always be cultured from patients with histologically proven IS. One study isolated spirochetes in only 50% of the cases (4). This finding suggests that culture alone may underestimate the true prevalence of the infection.
CMV Infection
The patient presented in this case report had serpiginous mucosal ulcers in his colon. Since IS tends not to cause mucosal ulceration, a search for other etiologies was undertaken. The CMV immunostain of the colonic biopsies was positive in the areas of inflammatory exudate and granulation tissue. This patient therefore had a concomitant CMV infection and IS. .
CMV belongs to the Herpes virus family and is a double stranded DNA virus. It has icosahedral symmetry; replicates in the nucleus and can cause both a lytic and latent infection. Viral replication within the cell cause large viral inclusions within the nucleus and occasional smaller inclusions in the cytoplasm, which leads to enlargement of the cells (cytomegaly). Disease due to CMV presents in patients with advanced AIDS, often with CD4 cell counts < 75/mm3 and viral loads > 100,000 copies/ml (23). .
The gastrointestinal tract is the second most common site of CMV disease in AIDS patients, following retinal involvement in frequency. Erosive inflammation due to CMV can occur in any portion of the GI tract. Patients with colonic involvement typically have symptoms such as diarrhea, bloody stools, and abdominal pain. Bowel perforation may occur, and GI involvement due to CMV is the commonest cause of acute surgical abdomen in AIDS. On colonoscopy, submucosal hemorrhage, erosions, and ulcerations are visualized. Severe erosions with pseudomembrane formation are occasionally reported.
Summary
Both CMV and IS likely contributed to the gastrointestinal symptoms described in this HIV positive patient, with the CMV colitis most likely responsible for the mucosal ulcerations, abdominal pain and weight loss, and IS contributing to the diarrhea. Our patient had a viral load of 644,000 copies/ml at the time of presentation, so he was above the cut off for susceptibility to CMV disease. Although not at increased risk for IS, patients with HIV/AIDS tend to be symptomatic with the infection.
IS can be a difficult histologic diagnosis to make and can be mistaken for a prominent brush border or mucin adherent to the mucosal surface. Since it can be inconspicuous, and since it can occur in asymptomatic adults, IS can be easily overlooked. Silver stains can enhance the detection and/or confirm the suspicion of intestinal spirochetes.
In an immunocompromised patient, the search for infectious agents should continue, even if one source of infection is detected.
REFERENCES
![]() Contributed by Akosua B Domfeh, MBchB and Alyssa Krasinskas MD
Contributed by Akosua B Domfeh, MBchB and Alyssa Krasinskas MD