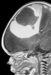
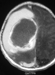
DIAGNOSIS: Chronic intracerebral hemorrhage of unknown etiology with reactive changes of the surrounding brain tissue and focal extramedullary erythropoiesis.
DISCUSSION:
The differential diagnosis of an intracranial mass lesion in a neonate includes neoplasm, infectious disease or hemorrhage.
In the first instance, we thought of a mass lesion due to a congenital supratentorial neoplasm, e.g., primitive neuroectodermal tumor, neuroblastoma, germ cell tumor, sarcoma or lymphoma. Using conventional stainings and immunohistochemistry, all these diagnoses could be excluded, particularly as no distinct neuroepithelial differentiation was found. Even more significantly, no hematopoietic stem cells (Figure 13) and only scattered B- and T-lymphocytes could be detected by immunohistochemistry. Furthermore, the peripheral blood cell count was normal.
The combination of dystrophic calcifications and hemorrhage was suggestive of infectious disease. The mother had been well throughout her pregnancy and major infectious diseases could be clinically excluded. Histologically and immunohistologically, we excluded congenital cerebral parasitic (cysticercosis, toxoplasmosis), viral (especially cytomegalovirus) and fungal infections.
In neonates, and especially in premature infants, cerebral hemorrhage remains the most common lesion. Formerly, intracranial hemorrhage was often due to erythroblastosis fetalis. The incidence of intracranial hemorrhages in neonates is high in fetomaternal autoimmunization (1). Using cranial sonography, Green et al., 1995 (2) demonstrated cerebral hemorrhage in three out of seven neonates with erythroblastosis fetalis. In the present case, the hemorrhage was first discovered in the 35th week of gestation. Clinically, erythroblastosis fetalis was excluded.
Very often, location of the hemorrhage provides clues as to its cause. Green and coworkers (2) showed that increased circulating nucleated erythrocytes in the newborn period can be markers of chronic fetal hypoxia, which, in turn, may be a risk factor for intracranial hemorrhage, especially periventricular hemorrhage (IVH). In this case, the hemorrhage was not localized periventricularly. On contrast-enhanced T1-weighted sagittal scan, MRI showed an extensive intracranial hemorrhage with a liquid area of high signal intensity and a central non-contrast-enhancing mass with a rim, also of high signal intensity (Fig. 15 and Fig. 16). This argues against asphyxia. Furthermore, we did not find any circulating nucleated erythrocytes in peripheral blood smears.
A differential diagnosis of the nests of cells in this case was extramedullary erythropoiesis, since erythroblasts have been described in encapsulated chronic subdural hematomas (3). Importantly, those cases did not demonstrate skull fractures or erythroblasts in peripheral blood smears. The authors suggested that these colonies of erythroblasts originate from the granulation tissue during the formation of neomembranes. In the present case, the hematoma was at least 3 weeks old. Accordingly, we found neomembranous brain parenchyma with reactive changes around the hemorrhage containing erythroblasts and nucleated red blood cells strongly reacting with an antibody against glycophorin A. Glycophorin A is a specific protein and the major red cell membrane sialoglycoprotein. Thus, as it is a lineage-specific marker, erythroblasts and more differentiated forms of the erythropoiesis, including erythrocytes, can be detected by the expression of glycophorin A.
In conclusion, to date, intracranial extramedullary erythropoiesis has only been described in chronic subdural hemorrhage. Here we present the first case of perifocal extramedullary erythropoiesis developed within the capsule of this hematoma, which was at least 3 weeks old and probably recurrent. The common denominator is intracranial bleeding with/without local hypoxia. One unresolved issue of this case is which cofactors are needed to induce intracranial erythropoiesis.
REFERENCES
![]() Contributed by Stefanie Scheil, Torsten Horn, Ulrich Kunz, Andreas Gabelmann, Anna-Leena Sirén, Rüdiger Klein, Christoph Schindler, Peter Möller, and Clemens Sommer
Contributed by Stefanie Scheil, Torsten Horn, Ulrich Kunz, Andreas Gabelmann, Anna-Leena Sirén, Rüdiger Klein, Christoph Schindler, Peter Möller, and Clemens Sommer