Final Diagnosis -- Pneumonia, Hilar Lymphadenitis and Sepsis Secondary to Rhodococcus equi
FINAL DIAGNOSIS
PNEUMONIA, HILAR LYMPHADENITIS, AND SEPSIS SECONDARY TO Rhodococcus equi.
Rhodococcus equi, originally identified by Magnusson as an agent capable of causing a granulomatous infection in horses in 1923,1,2 is an increasingly recognized opportunistic pathogen of humans. Since the initial description by Golub et al 3 initial description of R. equi infection in humans in 1967, more than 100 cases have been reported in the literature.2 While the organism primarily afflicts patients with impaired cellular immunity, cases have been described in patients with no known underlying predisposing condition.
R. equi is a water and soil organism which is most commonly found in environments associated with domesticated animals, whose manure provide required nutritional support for these organisms.4 Human infection is believed to occur secondary to the exposure of a susceptible individual to the appropriate environment. In the present case, the patient had a farm where he raised 27 horses which probaly account for his source of exposure. Once within the host, the bacterium becomes a facultative intracellular bacterium residing within macrophages and elicits an acute inflammatory response.
Clinically, the lung is the most frequent site of involvement, although other sites including the eyes, lymph nodes, soft-tissue, and bone have also been reported. 5 The patient typically presents with insidious onset of fever, chills, weight loss, dyspnea, and cough. Chest x-rays often reveal a thick-walled cavitating lower lobe pneumonia with hilar adenopathy, which in the typical clinical context of R. equi infections (i.e. impaired cellular immunity) mimics the Ghon complex of primary Mycobacterium tuberculosis infections. However, the presence of air fluid levels within the cavities and/or associated pleural effusions, which are frequently found with R. equi infections but rarely associated with M. tuberculosis, should raise the clinical suspicion of a R. equi infections.6
Due to the limited clinical experience with R. equi infections, treatment for such infections is empirically based. It has been observed that relapse occurs commonly. Therefore, long term (at least 2 months in duration) antibiotic treatment with repeated culture and sensitivity monitoring has been advocated.2,6 Most strains tested have been susceptible to glycopeptide antibiotics, rifampin, and macrolide antibiotics whereas the use of beta-lactam antibiotics, with the exception of imipenem, is not recommended since R. equi is often resistant. 2,6 In the current case, the patient was treated with a 2 month course of intravenous antibiotics (vancomycin and imipenem) followed by 4 months of oral antibiotics.
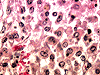 While the diagnosis of R. equi infections ultimately rests on culture techniques, characteristic tissue pathology, particularly in lung parenchyma, should raise the pathologist's suspicion of R. equi. The organism is an intracellular gram-positive coccobacillus which resides within foamy histiocytes within the infected host and is typically associated with a neutrophillic infiltrate and abundant bacteria during an acute infection. As the infection progresses, the foamy histiocytes accumulate lamellar calcified basophilic staining cytoplasmic bodies containing a central crystalline core surrounded by a less dense peripheral zone, Michaelis-Gutmann bodies (Image 19, not from current case, courtesy of Samuel A. Yousem, MD). This latter histologic picture, termed malakoplakia, is extremely rare within the lung and, when present, is highly suggestive of a R. equi infection.7
While the diagnosis of R. equi infections ultimately rests on culture techniques, characteristic tissue pathology, particularly in lung parenchyma, should raise the pathologist's suspicion of R. equi. The organism is an intracellular gram-positive coccobacillus which resides within foamy histiocytes within the infected host and is typically associated with a neutrophillic infiltrate and abundant bacteria during an acute infection. As the infection progresses, the foamy histiocytes accumulate lamellar calcified basophilic staining cytoplasmic bodies containing a central crystalline core surrounded by a less dense peripheral zone, Michaelis-Gutmann bodies (Image 19, not from current case, courtesy of Samuel A. Yousem, MD). This latter histologic picture, termed malakoplakia, is extremely rare within the lung and, when present, is highly suggestive of a R. equi infection.7
R. equi belongs to the order Actinomycetales, which includes several organism from which it must be differentiated from in a clinical microbiology laboratory including Nocardia and rapid growing Mycobacteriaceae.8,9,10 R. equi are non-fastidious intermittently gram-positive staining coccobacilli and filamentous forms with occasional fragmented and palisading forms, a morphology which may be confused with Corynebacterial organisms. R. equi may be partially acid fast, which may lead to confusion with Mycobacteriaceae. The organisms are slow growing on routine isolation agar (e.g. sheep blood) where the organism produces smooth and possibly mucoid white colonies which may produce a distinctive salmon-pink coloration with time. Colony morphology on tap water agar is very useful in differentiating members of the order Actinomycetales. Unlike Nocardia, Actinomadura, and Streptomyces, Rhodococcus lacks aerial hyphae. In addition, Rhodococcus exhibits rudimentary branching of its substrate hyphae which is useful in distinguishing it from rapid growing Mycobacteriaceae. R. equi is unable to hydrolyze casein, xanthine, and tyrosine, features which biochemically distinguish it from many members of Actinomycetales. R. equi is typically urease positive, a feature which is useful in distinguishing it from Corynebacterium. R. equi can be distinguished from other members of the Rhodococcus genus by its elaboration of equi factors which interact with the beta-toxin of Staphylococcus aureus, Listeria monocytogenes hemolysin, or phospholipase D of Corynebacterium pseudotuberculosis to form areas of complete hemolysis on sheep blood agar. Additional specialized testing, (ethylene glycol degradation, cell wall analysis, nucleic acid analysis) while available, is usually unnecessary to make the diagnosis of R. equi.
REFERENCES
- Magnuson H. Pneumonie beim Fohlen Ein neuer Eitererreger beim Pferd. Archiv fur Wissenschaftliche und Praktische Tierheilkunde. 1923;50:22-37.
- Linder R. Rhodococcus equi and Arcanobacterium haemolyticum: Two "Coryneform" bacteria increasingly recognized as agents of human infection. Emerg Infect Dis. 1997;3:145-153.
- Golub B, Falk G, Spink WW. Lung abscess due to Corynebacterium equi: report of first human infection. Ann Intern Med. 1967;66:1174-1177.
- Barton MD, Hughes KL. Ecology of Rhodococcus equi. Vet Microbiol. 1984;9:65-76.
- Scott MA, Graham BS, Verrall R, Dixon R, Schaffner W, Tham KT. Rhodococcus equi-An increasingly recognized opportunistic pathogen: Report of 12 cases and review of 65 cases in the literature. Amer J Clin Pathol 1995;103:649-655.
- Johnson DH, Cunha BA. Rhodococcus equi pneumonia. Sem Respir Infect. 1997;12:57-60.
- Kwon KY, Colby TV. Rhodococcus equi pneumonia and pulmonary malakoplakia in acquired immunodeficientcy syndrome: Pathologic features. Arch Pathol Lab Med. 1994;118:744-748.
- The aerobic gram-positive bacilli. In Color atlas and textbook of diagnostic microbiology. Eds Koneman EW, Allen SD, Janda WM, Schreckenberger PC, Winn WCJr. 1997; 5th ed. JB Lippencott Co, Philadelphia, pp 651-708.
- Beaman BL, Saubolle MA, Wallace RJ. Nocardia, Rhodococcus, Streptomyces, Oerskovia, and other aerobic Actinomycetes of medical importance. In Manual of clinical microbiology. Eds. Murray PR, Baron EJ, Pfaller MA, Tenover FC, Yolken RH. 1995; 6th ed. ASM press, Washington DC, pp 379-399.
- Staneck JL. Aerobic actinomycetes: Nocardia and related organisms. ASM teleconference. August 9,1994.
 Contributed by Scott M Kulich MD, PhD and William A Pasculle ScD
Contributed by Scott M Kulich MD, PhD and William A Pasculle ScD




 While the diagnosis of R. equi infections ultimately rests on culture techniques, characteristic tissue pathology, particularly in lung parenchyma, should raise the pathologist's suspicion of R. equi. The organism is an intracellular gram-positive coccobacillus which resides within foamy histiocytes within the infected host and is typically associated with a neutrophillic infiltrate and abundant bacteria during an acute infection. As the infection progresses, the foamy histiocytes accumulate lamellar calcified basophilic staining cytoplasmic bodies containing a central crystalline core surrounded by a less dense peripheral zone, Michaelis-Gutmann bodies (Image 19, not from current case, courtesy of Samuel A. Yousem, MD). This latter histologic picture, termed malakoplakia, is extremely rare within the lung and, when present, is highly suggestive of a R. equi infection.7
While the diagnosis of R. equi infections ultimately rests on culture techniques, characteristic tissue pathology, particularly in lung parenchyma, should raise the pathologist's suspicion of R. equi. The organism is an intracellular gram-positive coccobacillus which resides within foamy histiocytes within the infected host and is typically associated with a neutrophillic infiltrate and abundant bacteria during an acute infection. As the infection progresses, the foamy histiocytes accumulate lamellar calcified basophilic staining cytoplasmic bodies containing a central crystalline core surrounded by a less dense peripheral zone, Michaelis-Gutmann bodies (Image 19, not from current case, courtesy of Samuel A. Yousem, MD). This latter histologic picture, termed malakoplakia, is extremely rare within the lung and, when present, is highly suggestive of a R. equi infection.7