
![]() Contributed by Lara Berklite, MD and Sheldon Bastacky, MD
Contributed by Lara Berklite, MD and Sheldon Bastacky, MD
CLINICAL HISTORY
The patient was a man in his 20s with a 5 year history of deafness presenting with edema, nephrotic range proteinuria (urine protein / creatinine ratio of 15.5), mild hypertension (154/81 mm Hg) and severe anemia (hemoglobin-3.3 g/dL, hematocrit-9.7%). The patient had no history of diabetes mellitus, NSAIDs use, drugs or alcohol, or other listed medications and has no evidence of a gastrointestinal bleed. The patient did have a family history of deafness in both his mother and younger brother.
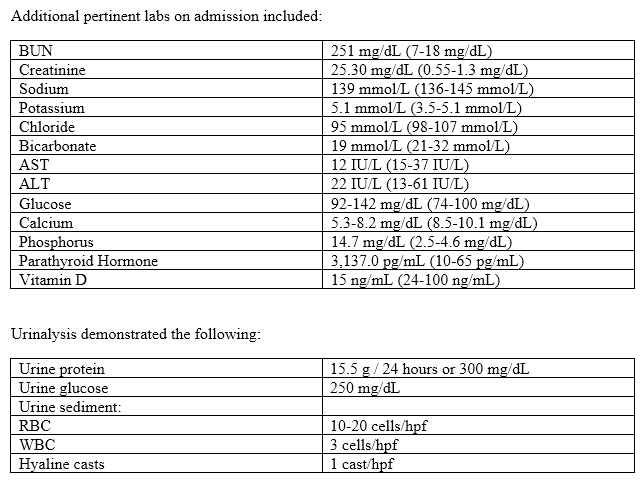
Anti-GBM antibody (<1.0 AI), HIV, and hepatitis B/C serologies were negative. Serum and urine immunofixation were negative for a monoclonal immunoglobulin. Serum complement levels were C4-22 mg/dL and C3-71 mg/dL (C3 reference range 90-180 mg/dL). The patient was noted to have small kidneys with cortical thinning bilaterally by ultrasound with the right and left kidneys measuring 7.1 cm and 7.5 cm in greatest dimension, respectively. A 1.8 cm cyst was identified on the upper pole of the left kidney. The patient received multiple units of packed red blood cells with an appropriate response in hematocrit and a percutaneous renal biopsy was performed.
GROSS EXAMINATION
Four cores of renal tissue measuring 1.0, 1.1, 1.1, and 1.4 cm in length, each measuring 0.1 cm in diameter were received. A 0.5 cm portion of the 1.1 cm core was submitted for immunofluorescence microscopy. A 0.4 cm portion of the 1.0 cm core was submitted for electron microscopy (Karnofsky's fixative). The remainder of the tissue was placed in the fresh formalin fixative and processed for light microscopy.
LIGHT MICROSCOPIC EXAMINATION
The tissue examined by light microscopy consisted of four cores of renal cortex and a small portion of renal medulla. The profiles of approximately 23 glomeruli were identified in the paraffin, frozen, and plastic sections, of which 11 (48%) were globally sclerotic. The non-obsolescent glomeruli had up to moderate mesangial proliferation and a few glomerular basement membrane double contours were present (Figure 1B, C, D). There was focal segmental glomerulosclerosis with focal epithelial cell proliferation involving 4/13 (31%) non-obsolescent glomeruli. There were no endocapillary proliferative lesions, necrotizing lesions, hyaline thrombi, glomerular basement membrane (GBM) spikes or GBM breaks. There was severe chronic tubular atrophy comprising greater than 90% of the cortical tubular profiles (Figure 1A). Some of the non-atrophic tubules showed evidence of acute tubular injury characterized by coarsely granular casts, degenerating cellular casts, and scattered hyaline casts. The interstitium was expanded by focal aggregates of foam cells, a moderate, predominantly small lymphocytic cellular infiltrate, and associated edema. Trichrome stain revealed moderate to severe established interstitial fibrosis. Approximately 17 small through medium-sized arteries were identified in the paraffin and plastic sections showing up to severe arterial sclerosis with fibroelastic intimal thickening and medial hypertrophy and arteriolar sclerosis. There was no evidence of vasculitis, thromboemboli, or thrombotic microangiopathy in the extraglomerular blood vessels examined.
IMMUNOFLUORESCENCE
The renal tissue examined by immunofluorescence microscopy consisted of one core of renal cortex and detached capsule containing 4 non-sclerotic glomeruli including one with focal segmental glomerulosclerosis, 1 globally sclerotic glomerulus, 2 arteries and 6 arterioles. Direct immunofluorescence is performed using a panel of 10 antisera. The findings included granular glomerular basement membrane and focal mesangial staining for C3 (1-2+), arterial and arteriolosclerosis with associated vascular staining for complement, and nonspecific staining of hyaline casts without light chain restriction.
ELECTRON MICROSCOPY
The ultrastructural findings were based on examination of one normocellular non-obsolescent glomerulus with multisegmental sclerosis visible on toluidine blue stained plastic section. The glomerulus showed extensive podocyte microvillous transformation, extensive foot process effacement, and focal lipoprotein resorption droplets within the podocytes. The glomerular basement membrane showed areas of thickening, splitting, and complex remodeling with a basket-weave pattern (Figure 2B, 2C). Measurements taken away from the remodeled glomerular basement membrane are thin with a range of 120 nm to 290 nm (mean +/- standard deviation = 192 +/- 48 nm) (normal adult male glomerular basement membrane thickness is 230-430 nm) (Figure 2A). Occasional circulating RBCs were seen within the glomerular capillaries. The tubules showed vacuolization and an RBC was imaged in the lumen. The tubular basement membranes exhibited thickening with a multilaminate, basket-weave pattern (Figure 2D). Three arteries were identified and showed fibroelastic thickening and arteriosclerosis. There was no ultrastructural evidence of a well-defined immune complex disorder or protein deposition disorder with organized substructure.
ANCILLARY STUDIES
Unstained frozen tissue sections were submitted to an outside institution for Type IV collagen isoform immunofluorescence evaluation. The evaluation revealed no detectable expression of alpha-5 Type IV collagen and redistribution of alpha-2 expression with accentuation in outer portions of glomerular capillary basement membranes. There was also absence of alpha-5 staining from tubular and Bowman's capsule basement membranes.