
![]() Contributed by Kevin Horn, MD and Darrell Triulzi, MD
Contributed by Kevin Horn, MD and Darrell Triulzi, MD
![]() Published on line in April 1997
Published on line in April 1997
PATIENT HISTORY:
The patient is a 67 year old female who presents to the hospital for a radical hysterectomy for endometrial carcinoma. The surgeons request a type and cross for four units of compatible packed red blood cells prior to surgery. The following results were obtained on crossmatch:
IMMUOHEMATOLOGICAL FINDINGS:
 FIGURE 1: Forward ABO typing of the patient's red blood cells (i.e., utilizing antibodies directed against the A and B antigens) demonstates 4+ activity against the A antigen, and no reactivity for B. Therefore, this patient "forwards" as a Type A. On reverse typing, the patient's serum is incubated with "test cells" of A and B type. The results show that the patient has 4+ reactivity in her serum against the B cells, and no reactivity directed against the A cells. Therefore, she has anti-B isohemagglutinins consistent with reverse typing as a group A strong antibody. Thus, she is shown to have Type A blood.
FIGURE 1: Forward ABO typing of the patient's red blood cells (i.e., utilizing antibodies directed against the A and B antigens) demonstates 4+ activity against the A antigen, and no reactivity for B. Therefore, this patient "forwards" as a Type A. On reverse typing, the patient's serum is incubated with "test cells" of A and B type. The results show that the patient has 4+ reactivity in her serum against the B cells, and no reactivity directed against the A cells. Therefore, she has anti-B isohemagglutinins consistent with reverse typing as a group A strong antibody. Thus, she is shown to have Type A blood.
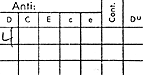 FIGURE 2: In the Rh typing, reagent containing antibodies against the "D" antigen are incubated with the patient's cells, and are shown her to have 4+ reactivity with her cells. Therefore, the patient's red blood cells express the D antigen and she would be classified as "Rh+". The D antigen is the most immunogenic antigen of the Rh system; anti-D antibodies can cause in vivo hemolysis and hemolytic disease of the newborn. Other Rh antigens, including C,c,E and e are not routinely typed unless the patient has a positive antibody screen. Note: There is no "d" antigen; this represents a "null allele." The far right column, Du, refers to a weak D expression. Du testing is a more sensitive method of detecting the D antigen in patients who initially type as Rh negative. Patients who type as "Du positive" are considered Rh positive.
FIGURE 2: In the Rh typing, reagent containing antibodies against the "D" antigen are incubated with the patient's cells, and are shown her to have 4+ reactivity with her cells. Therefore, the patient's red blood cells express the D antigen and she would be classified as "Rh+". The D antigen is the most immunogenic antigen of the Rh system; anti-D antibodies can cause in vivo hemolysis and hemolytic disease of the newborn. Other Rh antigens, including C,c,E and e are not routinely typed unless the patient has a positive antibody screen. Note: There is no "d" antigen; this represents a "null allele." The far right column, Du, refers to a weak D expression. Du testing is a more sensitive method of detecting the D antigen in patients who initially type as Rh negative. Patients who type as "Du positive" are considered Rh positive.
 FIGURE 3: The direct antiglobulin test (DAT) is performed to detect the presence of IgG antibody or complement (C3d), or both, on the surface of the red cell, using monoclonal antihuman globulin and complement. The presence of IgG or complement coating the red cell could be an indication of autoantibody produced by the patient against his/her own cells, antibodies produced by the patient coating transferred red cells, drug induced antibodies, or passive adsorption of IgG in patients with hypergammaglobulinemia or serum dysproteinemias of various causes. The DAT performed on our patient is negative.
FIGURE 3: The direct antiglobulin test (DAT) is performed to detect the presence of IgG antibody or complement (C3d), or both, on the surface of the red cell, using monoclonal antihuman globulin and complement. The presence of IgG or complement coating the red cell could be an indication of autoantibody produced by the patient against his/her own cells, antibodies produced by the patient coating transferred red cells, drug induced antibodies, or passive adsorption of IgG in patients with hypergammaglobulinemia or serum dysproteinemias of various causes. The DAT performed on our patient is negative.
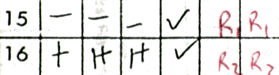 FIGURE 4: Many blood banks will mix the patient's serum with samples of two "screening cells". Screening cells are selected by the manufacturer to have representation of the major red blood cell antigens on one or both cells. Pathients with red cell antibodies would therefore react with the screening cells indicating the need for further evaluation. The reaction with screening cells takes place in three phases (represented by the three columns of results seen before the genotypes on the table in the image). The first column represents the initial mixing step of the patient serum sample and the screening cell. Known as the "immediate spin crossmatch," the specimens are mixed at room temperature, centrifuged, and examined for evidence of agglutination. The second column represents a warming phase in which the mixture is heated to approximate body temperature (about 37o C) and an enhancing meduium, such as LISS (low isotonic saline solution) is added to enhance antibody reactivity. The sample is incubated for ten minutes, recentrifuged and examined again for evidence of agglutination. The third and final column, the AGT or Coomb's phase, is performed using antihuman globulin which will promote agglutination of RBCs which are coated with IgG. As we can see here, the patient's serum showed no reactivity against screening cell I, but 1+ reactivity in all three columns for screening cell II. The fact that all three columns are positive suggests that the antibody is "cold-reactive" because it agglutinated cells at room temperature. "Cold reactive" antibodies are typically of the IgM class. The fact that the reaction is present in the Coomb's phase as well may indicate that an IgG antibody may be present as well.
FIGURE 4: Many blood banks will mix the patient's serum with samples of two "screening cells". Screening cells are selected by the manufacturer to have representation of the major red blood cell antigens on one or both cells. Pathients with red cell antibodies would therefore react with the screening cells indicating the need for further evaluation. The reaction with screening cells takes place in three phases (represented by the three columns of results seen before the genotypes on the table in the image). The first column represents the initial mixing step of the patient serum sample and the screening cell. Known as the "immediate spin crossmatch," the specimens are mixed at room temperature, centrifuged, and examined for evidence of agglutination. The second column represents a warming phase in which the mixture is heated to approximate body temperature (about 37o C) and an enhancing meduium, such as LISS (low isotonic saline solution) is added to enhance antibody reactivity. The sample is incubated for ten minutes, recentrifuged and examined again for evidence of agglutination. The third and final column, the AGT or Coomb's phase, is performed using antihuman globulin which will promote agglutination of RBCs which are coated with IgG. As we can see here, the patient's serum showed no reactivity against screening cell I, but 1+ reactivity in all three columns for screening cell II. The fact that all three columns are positive suggests that the antibody is "cold-reactive" because it agglutinated cells at room temperature. "Cold reactive" antibodies are typically of the IgM class. The fact that the reaction is present in the Coomb's phase as well may indicate that an IgG antibody may be present as well.
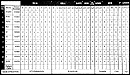 FIGURE 5: This figure shows the red cell panel; each row corresponds to a cell with a known phenotype of red cell antigens. By reacting patient serum with each of these cells in the three phases discussed above, one can determine the specificity of the patient's antibodies. Negative cells can be used to eliminate the presence of antibodies against antigens expressed on those cells. A "cross-out" method can be used by looking at the top of the table and crossing off antigens which are present in those cells. For example, in row #1, we see that the test cell is positive for D antigen. If our patient's serum does not react to this test cell, then we can surmise that she does not have anti-D, and we can cross D off of our list of potential causes of the reactivity. We can similarly eliminate the presence of antibodies against other antigens expressed on that cell. However, one must keep in mind that antibodies may show dosage and only cells exhibiting homozygous expression of an antigen can unequivocally rule out the presence of a corresponding antibody.
FIGURE 5: This figure shows the red cell panel; each row corresponds to a cell with a known phenotype of red cell antigens. By reacting patient serum with each of these cells in the three phases discussed above, one can determine the specificity of the patient's antibodies. Negative cells can be used to eliminate the presence of antibodies against antigens expressed on those cells. A "cross-out" method can be used by looking at the top of the table and crossing off antigens which are present in those cells. For example, in row #1, we see that the test cell is positive for D antigen. If our patient's serum does not react to this test cell, then we can surmise that she does not have anti-D, and we can cross D off of our list of potential causes of the reactivity. We can similarly eliminate the presence of antibodies against other antigens expressed on that cell. However, one must keep in mind that antibodies may show dosage and only cells exhibiting homozygous expression of an antigen can unequivocally rule out the presence of a corresponding antibody.
![]()
![]() FIGURES 6 and 7: This is our patient's pattern of reactivity to the panel in the last figure. We can also see, at the bottom, the row labelled "AC." The "autocontrol" is a mixture of the patient's serum with her own cells. If this row shows agglutination, then it implies that the patient has formed antibodies against her own cells. As we work our way down through the panel, we again notice that many rows show a similar pattern of reactivity to that seen in our screening cells, with positive reactions in all three phases. Seeing this, we are concerned about a the presence of a cold-reactive, or IgM, antibody. We should also think of an IgG antibody, which could be present in addition to the cold antibody. A quick method for determining the presence of an IgG antibody is to "prewarm" the sample before performing the AGT test; this prevents IgM antibodies from attaching to and causing agglutination of the red cells before the temperature reaches 37o C. As we can see in the prewarmed sample, the prior pattern of reactivity is no longer present, suggesting that the AGT reactivity was due to IgM antibody. Working our way through the anagram in figure 5 allows us to rule out all antibodies which are cold-reacting, except anti-Lea (the Lewis A antigen). Reactivity with the Lea positive cells indicates that the patient has antibodies directed against this antigen. Anti-Lea is rarely clinically significant (see the contributor's note below for a discussion).
FIGURES 6 and 7: This is our patient's pattern of reactivity to the panel in the last figure. We can also see, at the bottom, the row labelled "AC." The "autocontrol" is a mixture of the patient's serum with her own cells. If this row shows agglutination, then it implies that the patient has formed antibodies against her own cells. As we work our way down through the panel, we again notice that many rows show a similar pattern of reactivity to that seen in our screening cells, with positive reactions in all three phases. Seeing this, we are concerned about a the presence of a cold-reactive, or IgM, antibody. We should also think of an IgG antibody, which could be present in addition to the cold antibody. A quick method for determining the presence of an IgG antibody is to "prewarm" the sample before performing the AGT test; this prevents IgM antibodies from attaching to and causing agglutination of the red cells before the temperature reaches 37o C. As we can see in the prewarmed sample, the prior pattern of reactivity is no longer present, suggesting that the AGT reactivity was due to IgM antibody. Working our way through the anagram in figure 5 allows us to rule out all antibodies which are cold-reacting, except anti-Lea (the Lewis A antigen). Reactivity with the Lea positive cells indicates that the patient has antibodies directed against this antigen. Anti-Lea is rarely clinically significant (see the contributor's note below for a discussion).
 FIGURE 8: The four units (indicated by code numbers) which are to be crossmatched with our patient's serum are reacted in a similar fashion to our other panels. This serves the important purpose of making one last check to be sure there are no antibodies in the patient's serum against uncommon or variant antigens which were not represented on the usual panels. In this case, prewarming the sample allows us to eliminate the possibility of the patient reacting against Lewis a antigen which might be on the surface of these donor cells--since Lewis a antibodies are rarely clinically significant, most centers will transfuse units positive for this antigen, even if an antibody against this antigen is detected (as in this case).
FIGURE 8: The four units (indicated by code numbers) which are to be crossmatched with our patient's serum are reacted in a similar fashion to our other panels. This serves the important purpose of making one last check to be sure there are no antibodies in the patient's serum against uncommon or variant antigens which were not represented on the usual panels. In this case, prewarming the sample allows us to eliminate the possibility of the patient reacting against Lewis a antigen which might be on the surface of these donor cells--since Lewis a antibodies are rarely clinically significant, most centers will transfuse units positive for this antigen, even if an antibody against this antigen is detected (as in this case).