Brain Pathology Case of the Month - November 2011
 Contributed by Alejandro Ariel Gru1, Keith Fulling2, Arie Perry1,3
Contributed by Alejandro Ariel Gru1, Keith Fulling2, Arie Perry1,3
1Division of Neuropathology, Department of Pathology & Immunology, Washington University in St. Louis School of Medicine.
2Department of Pathology, St. John's Mercy. Saint Louis, MO.
3Department of Pathology, University of California in San Francisco.
CLINICAL HISTORY
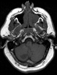
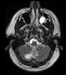
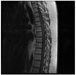
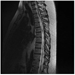
A 39 year-old-man with no significant past medical history presented with recurrent syncopal episodes, some of which were accompanied by nausea and vomiting. He also complained of recurrent headaches, gait problems and bilateral hand tremor. The physical exam confirmed the bilateral and symmetric intentional tremor and gait difficulties, without any motor or sensory deficits, including his cranial nerve examination. A brain MRI revealed a non-enhancing 2.4 x 1.7 cm mass within the right cerebellum; it was hypointense on T1 and hyperintense on T2 weighted sequences (Fig.1 and 2). Subtle leptomeningeal enhancement in the cortical sulci of both frontal lobes was suspicious for subarachnoid spread. Numerous T2 hyperintense lesions were also seen in the cervical and thoracic spine, the largest measuring 8 by 20 mm; these were similarly worrisome for CSF dissemination (Fig. 3 and 4). Providing further support for this notion, a cerebrospinal fluid specimen revealed scattered atypical small to medium sized cells with hyperchromatic nuclei, highly suspicious for malignant neoplasm. The patient subsequently underwent a subtotal resection of the cerebellar mass. Intra-operatively, the surgeon noted expanded cerebellar cortex with associated leptomeningeal coating or opacification suspicious for tumor involvement.
The patient subsequently underwent craniospinal irradiation and temozolomide chemotherapy. Two months later, profound pancytopenia was noted on a routine cell blood count (white blood count=0.4 K/mm3, hemoglobin=9.0 g/dl and platelets=42 K/ mm3). A CT scan showed sclerotic foci within the right sacrum, left iliac and left pubic bone. A bone marrow biopsy was done.
MICROSCOPIC PATHOLOGY
Histologically, there was a moderately to highly cellular neoplasm that extensively invaded the cerebellar cortex (Fig. 5) and, to a lesser extent, the underlying white matter, often entrapping both internal granular layer and Purkinje cell neurons, the latter involved in occasional vague perineuronal satellitosis. The tumor cells showed mild nuclear pleomorphism, with the majority of them containing round to oval nuclei with delicate chromatin and occasional clear perinuclear halos (Fig. 6). Rare mitotic figures were seen, but there was no microvascular proliferation or necrosis. The tumor was also rich in basophilic mucin (Fig. 7), confirmed by an Alcian blue stain, and had a microcystic growth pattern. No definitive rosettes, perivascular pseudorosettes, Rosenthal fibers, eosinophilic granular bodies, or regions of lipidization were seen. A bone marrow biopsy also revealed aggregates of malignant cells embedded within a fibrillary to collagenous matrix with tumor cells resembling those encountered previously in the cerebellum (Fig. 8 and 9).

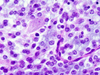


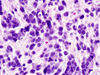



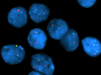
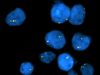


By immunohistochemistry, the tumor cells were strongly positive for S-100 protein (Fig. 10), and minor subsets were positive for glial fibrillary acidic protein (GFAP; Fig. 11) and synaptophysin. A fraction of tumor cells also displayed cytoplasmic WT1 positivity (Fig. 12). Both GFAP and WT1 stains highlighted thin perinuclear rims and tadpole-like processes in the tumor cells. The Ki-67 labeling index was low to moderate, focally reaching up to 6.4%. The tumor cells were negative for neurofilament protein and Neu-N, although the latter highlighted entrapped internal granular cell neurons. FISH was performed on paraffin-embedded tissue with probes localizing to 1p32, 1q42, 19p13 and 19q13 as previously described2. Deletions of both 1p and 19q were identified (Fig. 13 and 14). There was also evidence of 1q gain and monosomy 19 in a subset of tumor cells. Immunohistochemistry performed in the bone marrow biopsy showed that the tumor cells were also positive for S100 (Fig 15), synaptophysin, WT-1 (Fig. 16) and CD56, but negative for GFAP, CAM5.2, HMB-45 pancytokeratin, and myeloperoxidase.
FINAL DIAGNOSIS
















 International Society of Neuropathology
International Society of Neuropathology