Brain Pathology Case of the Month - January 2006
 Contributed by Fabrice Chrétien MD, PhD1, Jérôme Servan MD2, Jacqueline Mikol MD, PhD3, Michčle Trierweiller MD2, Dominique Elghozi MD2, Françoise Gray MD, PhD3
Contributed by Fabrice Chrétien MD, PhD1, Jérôme Servan MD2, Jacqueline Mikol MD, PhD3, Michčle Trierweiller MD2, Dominique Elghozi MD2, Françoise Gray MD, PhD3
1. INSERM EMI 0011, Université Paris XII, Créteil, France. & Service d'Histologie - Hôpital Henri Mondor AP-HP, 94010 Créteil cedex, France.
2. Service de Neurologie, Hôpital René Dubos, Pontoise, France
3. Service Central d'Anatomie et de Cytologie Pathologiques, Hôpital Lariboisičre AP-HP, Paris, France
 Published on line in January 2006
Published on line in January 2006
CLINICAL HISTORY:
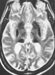
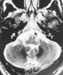
A 70 year-old man presented with a 8 year history of cognitive decline. Initial presenting signs included mild memory loss, behavioural disturbances, and transient episodes of disorientation. Neurological examination revealed frontal release signs, including severe confusion, hallucinations and bilateral grasping, pyramidal signs, facial dyskinesia and dysmetria. During this first hospitalization, his "mini mental status" (MMS) was quoted as 25/30. Laboratory investigations showed undetectable ceruloplasmin in serum, increased ferritin (1777 µg/L - normal 30-300) and a slight decrease of serum iron (11,8µmol/l -normal 12-30) and copper (0.06 mg/l - normal 0.8-1.4) levels. Fundus oculi was normal; there was neither Kayser-Fleischer ring nor retinal abnormality. There were no signs of diabetes mellitus. Brain MRI revealed bilateral increased signal intensities of deep nuclei on T1 weighted that were more marked on T2 weighted sequences. The abnormalities involved predominantly the putamen, caudate nucleus, pulvinar, and dentate nucleus. Contrast infusion did not show any signal modification. (Figures 1 and 2). Due to a suspicion of hemochromatosis, a liver biopsy was performed. It showed normal liver architecture, absence of cirrhosis and marked iron deposition. Aspirated bone marrow showed normal cytology and cellular count but massive iron deposition.
The patient's condition progressively worsened. Six years after admission, he was bedridden with obvious dementia (MMS less than 5/30) and mutism. He died from aspiration bronchopneumonia, 8 years after the onset of neurological signs.
There was neither a family history of similar disease nor consanguinity between the close parents. Two brothers had diabetes mellitus, progressive mental deterioration, movement disorders and cerebellar ataxia, together with aceruloplasminemia whereas serum ferritin and serum iron were increased. In the descendants, two of the three children presented with a low blood level of ceruloplasmin compatible with an heterozygous form of the disease.
POSTMORTEM EXAMINATION:
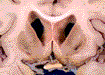

Visceral examination showed diffuse hemosiderosis of the heart, liver and lung.
Gross examination of the brain (1170g) showed predominant involvement of the basal ganglia with rust-brown discoloration and cavitation of the striatum, posterior thalamus and dentate nuclei (Figures 3 and 4). It was associated with mild cortico-subcortical atrophy. The cerebral and cerebellar cortex, white matter, substantia nigra and globus pallidus were otherwise macroscopically normal.
MICROSCOPIC DESCRIPTION:

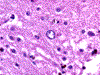

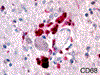

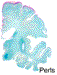

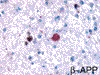


Prominent changes were observed in the striatum, posterior thalamus and dentate nucleus. Lesions were symmetrical and bilateral. In the necrotic foci, there was severe neuronal loss with fibrosis (Figure 5) associated with marked astrocytosis and macrophagic activation better identified on GFAP, HLA-DR and CD68 immunostains. Astrocytosis included gemistocytic astrocytes and Alzheimer's type II glia (Figures 6 and 7). Macrophages expressing CD68 and HLA-DR were numerous and some of them had the morphological characteristics of Opalski cells (Figures 8 and 9). Iron deposits better identified on Perls stain were abundant in the necrotic foci but were more diffuse, and also found in the cerebral and cerebellar cortex (Figures 10 and 11) and to a lesser extent in the white matter. They were observed in the cytoplasm of macrophages and astrocytes but were also present in neurons or free in the neuropil. Axonal swellings better demonstrated by Bodian silver impregnation or beta-Amyloid Protein Precursor (APP) immunostaining (Figure 12), were numerous within and around the necrotic foci. They frequently contained iron deposits.
There was severe involvement of the cerebellar cortex with iron deposition, loss of Purkinje cells and proliferation of Bergmann glia (Figure 13). The white matter, globus pallidus and substantia nigra did not show significant changes except slight iron deposits (Figure 14).
FINAL DIAGNOSIS













 International Society of Neuropathology
International Society of Neuropathology