
![]() Contributed by Isil Z Yildiz, MD and Lydia C. Contis, MD
Contributed by Isil Z Yildiz, MD and Lydia C. Contis, MD
CLINICAL HISTORY
Patient is a 46-year-old female with no significant past medical history. She presented to an outside hospital with a three day history of shortness of breath and palpitations. A chest x-ray revealed bilateral lung infiltrates. She was subsequently intubated for respiratory failure and sedated. She also had black, tarry stools and bloody orogastric tube output, and was subsequently admitted to an outside hospital intensive care unit for sepsis. Reported initial complete blood count values included a hemoglobin value of 7.4 gm/dl, platelet count of 13x10E+9/L, and white blood cell count of 10.3x10E+9/L, The peripheral blood differential reportedly demonstrated 15% blasts. She was transferred to the intensive care unit at UPMC for further management. A bone marrow biopsy was performed for further evaluation.
Peripheral Blood:
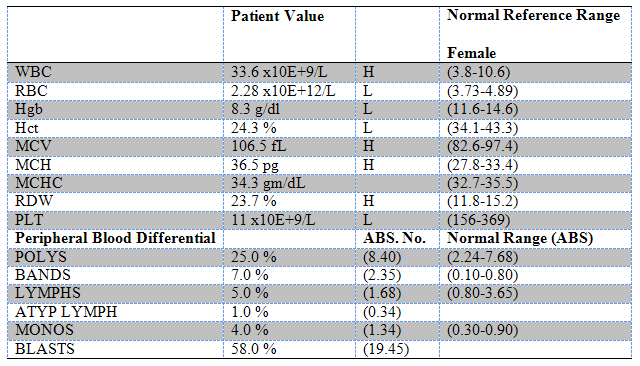
The results of the complete blood count (CBC) are illustrated in Table 1. These demonstrate an elevated white blood cell count with 58% blasts along with a macrocytic anemia and thrombocytopenia.
Table 1: CBC Data:
The peripheral blood smear demonstrated small and intermediate size blasts with basophilic or salmon colored cytoplasm , prominent hofs, fine or dense cytoplasmic granules and rare Auer rods. Dysplastic neutrophils were also noted (Figure 1 and Figure 2).
Figure 1: Small and intermediate size blasts with prominent hofs (arrows), fine or dense cytoplasmic granules and Auer rods (arrowhead) (Wright-Giemsa stain,x1000).
Figure 2: Small and intermediate size blasts with basophilic or salmon colored cytoplasm (black arrows) and a dysplastic neutrophil (red arrow) (Wright-Giemsa stain,x1000).
Bone Marrow:
The bone marrow differential demonstrated numerous blasts (50% by manual differential of 300 cells) along with complete maturation of myeloid cells (Table 2). The blasts demonstrated morphologic features similar to those seen in peripheral blood (Figure 3). The bone marrow biopsy was normocellular for age (50-60% cellular) (Figure 4 and Figure 5).
Table 2: Bone marrow differential:
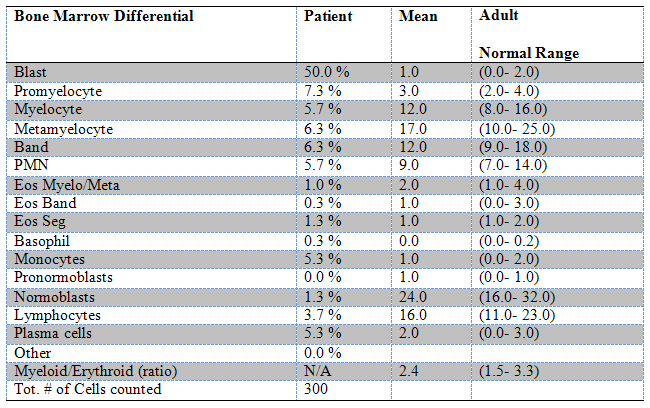
Figure 3: Bone marrow aspirate smear with many blasts containing granules (Wright-Giemsa stain,x1000).
Figure 4: Bone marrow core biopsy (Hematoxylin-eosin,x100 and x400).
Figure 5: Bone marrow core biopsy (Hematoxylin-eosin,x400).
Immunuhistochemical Stains:
CD56, CD68, and CD34 immunohistochemical stains performed on the biopsy each demonstrated few scattered positive cells, whereas CD117 showed rare positive cells. PAX5 was negative in the blasts.
Cytochemical Stains:
A chloroacetate esterase stain was performed on the bone marrow aspirate which demonstrated that many of the granulated cells were strongly positive (Figure 6).
Figure 6: Chloroacetate esterase stain (x1000).
Flow Cytometry:
Flow cytometric immunophenotypic studies performed on the bone marrow demonstrated an immature myeloid population comprising approximately 47% of the total events with the following phenotype:
There was also a second small population of myeloblasts comprising approximately 3.4% of the total events with the following phenotype:
Figure 7: Selected flow cytometry histograms: A; demonstrates 4.4% dim CD45+ cells (pink) , 2.9% bright CD45+ cells (blue), and 3.5% CD34+ cells (orange), B; demonstrates CD13/33+ and CD56+ immature myeloid population (orange).
Fluorescence In-Situ Hybridization Studies:
FISH analysis was positive for the RUNX1T1/RUNX1 gene rearrangement in 196 of the 201 interphase cells examined (97.5%) (Figure 8). FISH analysis was negative for the PML/RARA gene rearrangement in all of the interphase cells examined in the specimen.
Figure 8: Fluorescence in situ hybridization performed using the Vysis DNA probes (Abbott Molecular Inc., Des plaines, IL).
Classical cytogenetics demonstrated the presence of a t(8;21)(q22;q22), inv(9)(p11q13) . The analysis revealed an abnormal female bone marrow chromosome with a pericentric inversion of one chromosome 9 (Figure 9, red arrow) along with a translocation between the long arms of chromosomes 8 and 21 (Figure 9,black arrows). The inv(9)(p11q13) chromosome occurs in about 2% of the population and is considered to be a normal variant.
Figure 9: Classical cytogenetic analysis. 20 trypsin-Giemsa banded metaphase cells were analyzed. All 20 cells had a 46, XX, t(8;21)(q22;q22),inv(9)(p11q13) chromosome pattern.