


FINAL DIAGNOSIS
PAPILLARY THYROID CARCINOMA, FOLLICULAR VARIANT (0.5 CM), ENCAPSULATED AND LOCATED IN THE RIGHT LOBE. THERE IS NO INVASION OF THE TUMOR CAPSULE.
MOLECULAR ANATOMIC PATHOLOGY TESTING
Molecular anatomic pathology testing was performed. Realtime PCR for RET/PTC1, RET/PTC3, and PAX8/PPARγ rearrangements were performed, with negative results (Figure 6, Realtime PCR for chromosomal rearrangements). Post-PCR melting curve analysis for NRAS codon 61, HRAS codon 61, and KRAS codons 12/13 demonstrated peaks at melting points consistent with the wild type genotype of each negative control. Analysis for the BRAF V600E mutation, showed a mild shift in melting temperature, however (Figure 7, Post-PCR melting curve analysis for RAS and BRAF mutations). PCR amplification and Sanger sequencing was therefore performed. On agarose gel electrophoresis, a larger PCR product, in addition to the expected target, was seen (Figure 8, Gel electrophoresis of BRAF PCR product (arrow: larger product containing insertion)). Sanger sequencing demonstrated an insertion/duplication of 51 nucleotides (17 amino acids): p.A599insIFLHEDLTVLIGDPGLAA (Figure 9, Sanger sequencing showing insertion/duplication: p.A599insIFLHEDLTVLIGDPGLAA).
DISCUSSION
Driver mutational events in papillary thyroid carcinoma (PTC) include several that result in constitutive activation of the mitogen activated protein kinase (MAPK) pathway. These include chromosomal rearrangements affecting the RET gene (most commonly with H4 and ELE1 genes in the case of RET/PTC1 and RET/PTC3 rearrangements) as well as point mutations at NRAS codon 61, HRAS codon 61 and KRAS codon 12/13. The most prevalent mutational event, however, occurring in approximately 40-45% of PTC, is point mutation of BRAF (1, 2).
The great majority of BRAF mutations involve a T to A transversion at nucleotide 1799, resulting in the valine to glutamate substitution at residue 600 (V600E). Rarely, point mutations occur at other nucleotides. The most common of these is the K601E substitution, comprising approximately 1% of BRAF mutated PTC (1). Additionally, a rare chromosomal rearrangement of BRAF with AKAP9 has been identified, most commonly in radiation-related PTC developing shortly (e.g. within 10 years) after exposure (3).
BRAF V600E mutation is most commonly found in classic papillary carcinoma and tall cell variant. Additionally, BRAF mutation is associated with a more aggressive tumor phenotype, including higher rates of extrathyroidal extension, lymph node and distant metastasis, and recurrence (1).
Other rare alterations of the BRAF gene include insertions and/or deletions. The case described above is quite unusual as, to date, the largest reported BRAF insertion consisted of 18 nucleotides, 6 amino acids (4). Table 1 summarizes reported BRAF insertions in thyroid carcinoma. The clinical significance of these insertions remain largely unknown.
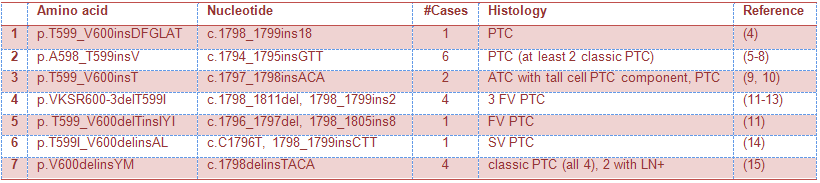
Table 1: BRAF insertions reported in thyroid carcinoma
ATC-anaplastic thyroid carcinoma, FV-follicular variant, SV-solid variant, LN-lymph node
Molecular testing for BRAF, NRAS, HRAS and KRAS mutation can be performed through LightCycler PCR followed by melting curve analysis using fluorescence resonance energy transfer (FRET) probes. These probes are not degraded during PCR, such that they can be used in post-PCR melting curve analysis. A single mismatch between the sequence of the amplified product and the designed probe will result in a change in the product's melting temperature and a shift in the melting peak compared to a wild type control (1). Testing for known mutational events in PTC has become particularly useful in the evaluation of thyroid fine needle aspirate (FNA) specimens. One hundred percent of nodules found to harbor a BRAF V600E mutation are malignant. RET/PTC and PAX8/PPARγ translocations also indicate 100% probability of malignancy, and 86% in cases with RAS mutation, greatly improving the diagnostic utility of thyroid FNA. Approximately 30% of sporadic PTC do not harbor any of these known mutational events, however, such that negative results on molecular anatomic pathology testing cannot rule out PTC (2).
REFERENCES
![]() Contributed by Rebecca Leeman-Neill, MD, PhD, Somak Roy, MD, and Yuri Nikiforov, MD, PhD
Contributed by Rebecca Leeman-Neill, MD, PhD, Somak Roy, MD, and Yuri Nikiforov, MD, PhD